1. Introduction
Hyperuricemia, broadly defined as an elevation in serum uric acid beyond physiological solubility thresholds, is increasingly recognized as more than a simple biochemical abnormality. It is, of course, the central pathogenic driver of gout, but its clinical significance extends well beyond acute arthritis alone. Persistently elevated urate levels have also been associated with chronic kidney disease, hypertension, metabolic syndrome, insulin resistance, and broader cardiometabolic dysfunction (Borghi et al., 2014; Dalbeth et al., 2016; Johnson et al., 2013). In many patients, hyperuricemia remains clinically silent for years, yet the underlying biochemical environment continues to favor monosodium urate crystal deposition, low-grade inflammation, and progressive metabolic disturbance. That silent progression is precisely what makes the condition both deceptively common and, in some cases, therapeutically frustrating.
Conventional urate-lowering therapy remains the cornerstone of clinical management. Agents such as allopurinol and febuxostat, primarily through xanthine oxidase inhibition, are effective in reducing urate production and preventing long-term crystal burden (Pacher et al., 2006; Richette et al., 2017). Still, the real-world management of hyperuricemia is often less straightforward than treatment guidelines may suggest. Some patients experience adverse effects, hypersensitivity reactions, gastrointestinal intolerance, or poor long-term adherence, while others remain reluctant to initiate chronic pharmacotherapy for what is initially perceived as an asymptomatic laboratory abnormality (Stamp & Chapman, 2013). In practice, this creates a persistent therapeutic gap—particularly for individuals seeking gentler, adjunctive, or more “natural” approaches that might support urate control before recurrent gout or organ-level complications become established.
It is within this gap that botanical and phytopharmaceutical interventions have drawn growing attention. Not all plant-based products, admittedly, are scientifically persuasive, and many suffer from weak standardization or exaggerated health claims. However, some medicinal plants have accumulated a more credible mechanistic foundation and may warrant serious clinical exploration. Among these, Orthosiphon stamineus Benth., commonly known as cat’s whiskers or Java tea, has emerged as a particularly interesting candidate. Traditionally used in Southeast Asian medicine for urinary, renal, and metabolic complaints, O. stamineus has been investigated for a range of biological activities, including antioxidant, anti-inflammatory, nephroprotective, and metabolic effects (Ameer et al., 2012; Yam et al., 2009).
From a urate-lowering perspective, the pharmacologic rationale is especially compelling. Preclinical studies suggest that O. stamineus and its polyphenolic constituents may influence serum uric acid through several complementary mechanisms rather than a single isolated pathway. These include possible inhibition of xanthine oxidase, enhancement of renal urate excretion, and modulation of inflammatory and oxidative pathways that are increasingly understood to interact with urate metabolism and gout pathophysiology (Cos et al., 1998; Kong et al., 2000; Lin et al., 2002). This “polymolecular” profile may be particularly relevant in metabolic disorders, where disease expression rarely arises from one pathway alone.
A major contributor to this therapeutic interest lies in the phytochemical composition of the plant. Standardized extracts of O. stamineus commonly contain rosmarinic acid, sinensetin, eupatorin, and related methoxylated flavones, compounds with documented antioxidant and anti-inflammatory properties (Akowuah et al., 2004; Damsud et al., 2014; Petersen & Simmonds, 2003). Rosmarinic acid, for instance, has been widely studied for its ability to attenuate oxidative stress and inflammatory mediator activity, both of which are relevant to urate-induced tissue injury and gout flare biology (Al-Dhabi et al., 2020). Likewise, flavonoid compounds such as sinensetin and eupatorin have shown biochemical activities that plausibly intersect with urate metabolism and inflammatory signaling. Taken together, these constituents suggest that O. stamineus may not simply act as a “traditional herb,” but rather as a standardized botanical with mechanistic relevance to hyperuricemia.
Yet despite this promising preclinical and phytochemical foundation, the clinical evidence base remains surprisingly limited. There is still a noticeable gap between laboratory plausibility and documented human outcome data, particularly in real-world settings. That gap matters. Mechanistic promise alone is not enough; what ultimately matters is whether measurable biochemical improvement can be observed in patients under routine use conditions. Even small observational datasets, when carefully reported and appropriately interpreted, can provide useful preliminary insight and help justify more rigorous prospective investigation.
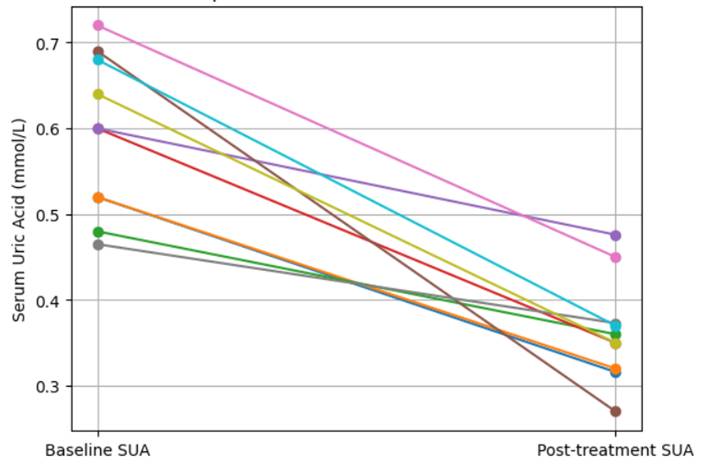
Against this background, the present retrospective case series was undertaken to describe changes in serum uric acid levels among adults with laboratory-confirmed hyperuricemia who consumed Canssu5®, a standardized Orthosiphon stamineus extract formulated to contain approximately 6% rosmarinic acid. Rather than attempting to make definitive therapeutic claims, this report aims to provide early clinical observations on a standardized botanical intervention that may hold translational relevance in the management of hyperuricemia. In doing so, it seeks to contribute a modest but clinically meaningful piece of evidence to an area where patient interest is growing, yet formal human data remain scarce.