1. Introduction
Acne vulgaris (AV) is often discussed as a routine dermatological condition, yet its biological and social dimensions make it far more complex than the occasional inflammatory lesion suggests. At its core, AV is a chronic disorder of the pilosebaceous unit, involving interactions among the hair follicle, sebaceous gland, keratinocytes, and immune mediators. These interactions unfold gradually and often persist for years, particularly during adolescence and early adulthood (Tuchayi et al., 2015). Epidemiological estimates indicate that acne affects a striking proportion of the global population—roughly 85% of adolescents and a notable fraction of adults—making it one of the most common inflammatory skin disorders worldwide (Heng & Chew, 2020). The clinical manifestations of acne may appear superficial, yet their implications extend beyond the skin. Persistent lesions, scarring, and treatment-resistant inflammation frequently coincide with reduced self-esteem and psychological distress, illustrating that AV should be understood as both a dermatological and psychosocial challenge.
The biological mechanisms underlying acne are multifaceted and still evolving in scientific understanding. Traditionally, pathogenesis has been framed around four interacting processes: increased sebum production, follicular hyperkeratinization, inflammation, and microbial colonization. While this framework remains useful, recent research suggests that microbial ecology within the skin may be particularly central to disease progression (Platsidaki et al., 2018). The pilosebaceous unit hosts a dynamic microbial community dominated by Cutibacterium acnes, a Gram-positive anaerobic bacterium that typically behaves as a benign commensal. Under stable conditions, this microorganism contributes to maintaining the skin’s ecological balance. However, shifts in environmental conditions within the follicle—changes in sebum composition, oxygen availability, or host immune responses—can encourage certain C. acnes phylotypes to adopt a more inflammatory phenotype (Gannesen et al., 2019; de Sousa et al., 2020).
Increasingly, researchers are recognizing that acne cannot be reduced to the activity of a single microbial species. Instead, the condition appears to reflect a broader disturbance in the skin microbiome. When microbial diversity declines, competitive balance among commensal organisms can shift, allowing opportunistic species such as Staphylococcus aureus or Staphylococcus epidermidis to exert a stronger influence on the inflammatory environment (Fournière et al., 2020). Interactions among these microorganisms are not merely passive; they involve chemical communication, metabolic cooperation, and the formation of complex biofilm structures. Biofilms, in particular, have attracted attention because they provide microorganisms with protection against host defenses and antimicrobial agents, thereby sustaining chronic inflammation (Brandwein et al., 2016). Seen from this perspective, acne emerges less as a simple infection and more as a form of microbial dysbiosis in which ecological equilibrium has been disrupted.
Despite these advances in understanding microbial dynamics, therapeutic approaches have changed only gradually. Conventional treatments remain dominated by topical and systemic antibiotics, retinoids, and hormonal interventions. These therapies can be effective in many patients, yet their use is not without complications. Antibiotics, for instance, have long been a cornerstone of acne management, but prolonged exposure has contributed to the emergence of antibiotic-resistant bacterial strains within the skin microbiota (Farrah & Tan, 2016). The implications of this resistance extend beyond dermatology. Antimicrobial resistance has become a global public health concern, with projections suggesting that drug-resistant infections could impose significant health and economic burdens in the coming decades (O’Neill et al., 2016). Compounding this problem is the relatively limited pipeline of new antimicrobial agents. Global health authorities have warned that the development of innovative antibiotics has not kept pace with the accelerating spread of resistance (WHO, 2021). Within this context, dermatological conditions such as acne—where long-term antibiotic therapy is common—highlight the urgent need for alternative therapeutic strategies.
One possible avenue lies in the exploration of natural products, which historically have played a central role in antimicrobial drug discovery. Before the modern era of synthetic pharmaceuticals, a large proportion of therapeutic agents were derived from naturally occurring compounds produced by plants, fungi, and microorganisms (Pham et al., 2019). Among microbial producers, members of the phylum Actinobacteria have proven particularly prolific. Species belonging to the order Actinomycetales, especially those within the genus Streptomyces, are responsible for the discovery of numerous clinically important antibiotics and other bioactive metabolites. Their capacity to generate structurally diverse secondary metabolites has long attracted interest from pharmacologists and microbiologists alike.
Yet the terrestrial environments from which most actinobacteria were originally isolated have been intensively studied for decades. As a consequence, researchers frequently encounter previously characterized strains or rediscover known compounds, slowing the pace of novel drug discovery. In response to this challenge, scientific attention has gradually shifted toward marine ecosystems, which remain comparatively underexplored. Marine habitats—particularly those associated with sediments, sponges, and other invertebrates—are increasingly recognized as reservoirs of microbial diversity and chemical novelty (Santos et al., 2019). Organisms inhabiting these environments experience distinctive ecological pressures, including fluctuating salinity, limited nutrients, and varying oxygen concentrations. Such conditions may drive the evolution of unique biosynthetic pathways and unusual secondary metabolites (Gavriilidou et al., 2021).
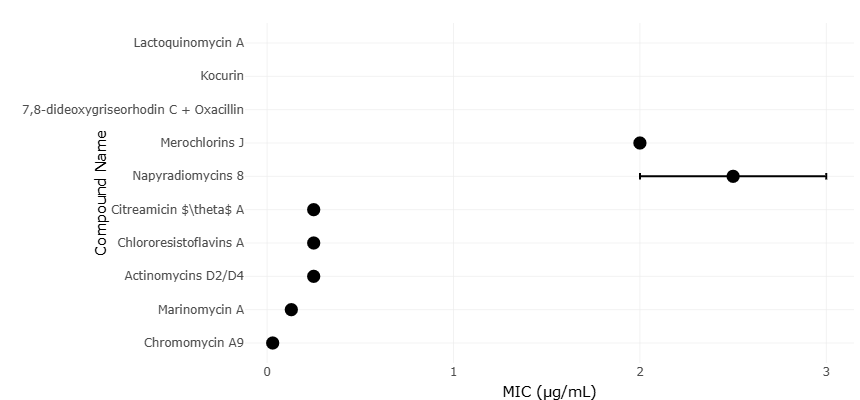
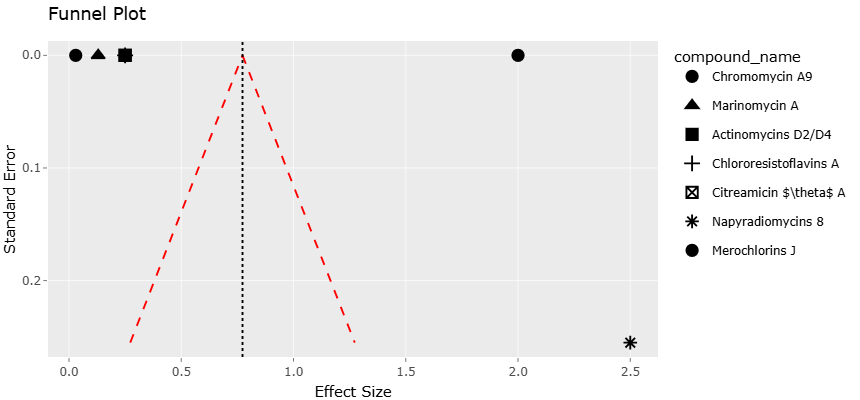
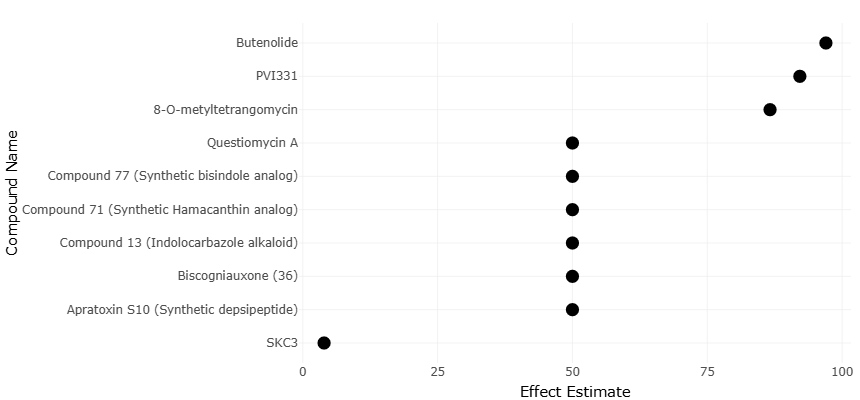
Indeed, marine actinobacteria have already yielded a variety of structurally distinctive compounds with antibacterial and anti-infective activity. Polyketide metabolites, for example, have demonstrated considerable chemical diversity and biological potency (Arasu et al., 2013). Other compounds—such as marinopyrroles, napyradiomycins, and meroterpenoids—illustrate the remarkable biosynthetic versatility of marine microorganisms and their potential pharmaceutical relevance (Cheng et al., 2013; Hughes et al., 2008). Additional metabolites derived from marine actinomycetes have shown activity against clinically important pathogens, including methicillin-resistant Staphylococcus aureus (Paderog et al., 2020). These discoveries collectively suggest that marine microorganisms may represent a largely untapped resource for antimicrobial development.
Interest in these organisms has been further strengthened by advances in microbial cultivation strategies. Co-culture techniques, for instance, allow different microbial species to grow together, often triggering the production of previously silent biosynthetic pathways. Such interactions can lead to the emergence of novel metabolites that remain undetected under standard laboratory conditions (Yu et al., 2019). Similarly, experimental approaches involving microbial signaling molecules and environmental stressors have been used to stimulate metabolite production in marine actinomycetes (Sung et al., 2017). These methodological innovations hint at a broader landscape of chemical diversity that has yet to be fully explored.
Within dermatological research, the potential of marine actinobacteria is only beginning to receive systematic attention. A number of studies have reported antibacterial compounds derived from marine actinomycetes that exhibit activity against pathogens associated with skin infections. In some cases, these metabolites interfere with bacterial communication systems or disrupt biofilm formation, suggesting mechanisms of action that differ from traditional antibiotics (Dholakiya et al., 2017). Such properties are particularly relevant to acne, where microbial signaling and biofilm development contribute to inflammation and treatment resistance.
Nevertheless, despite the growing interest in marine microbial metabolites, their direct application to acne-associated bacteria remains relatively underexplored. Systematic evaluation of available literature indicates that most studies have focused on activity against Staphylococcus species, while comparatively few investigations have examined effects on Cutibacterium acnes itself (de La Hoz-Romo et al., 2022). This imbalance raises intriguing questions. It may reflect methodological challenges associated with cultivating anaerobic bacteria, or perhaps a historical emphasis on more easily studied pathogens. Whatever the reason, the gap suggests that the therapeutic potential of marine actinobacteria for acne treatment has not yet been fully realized.
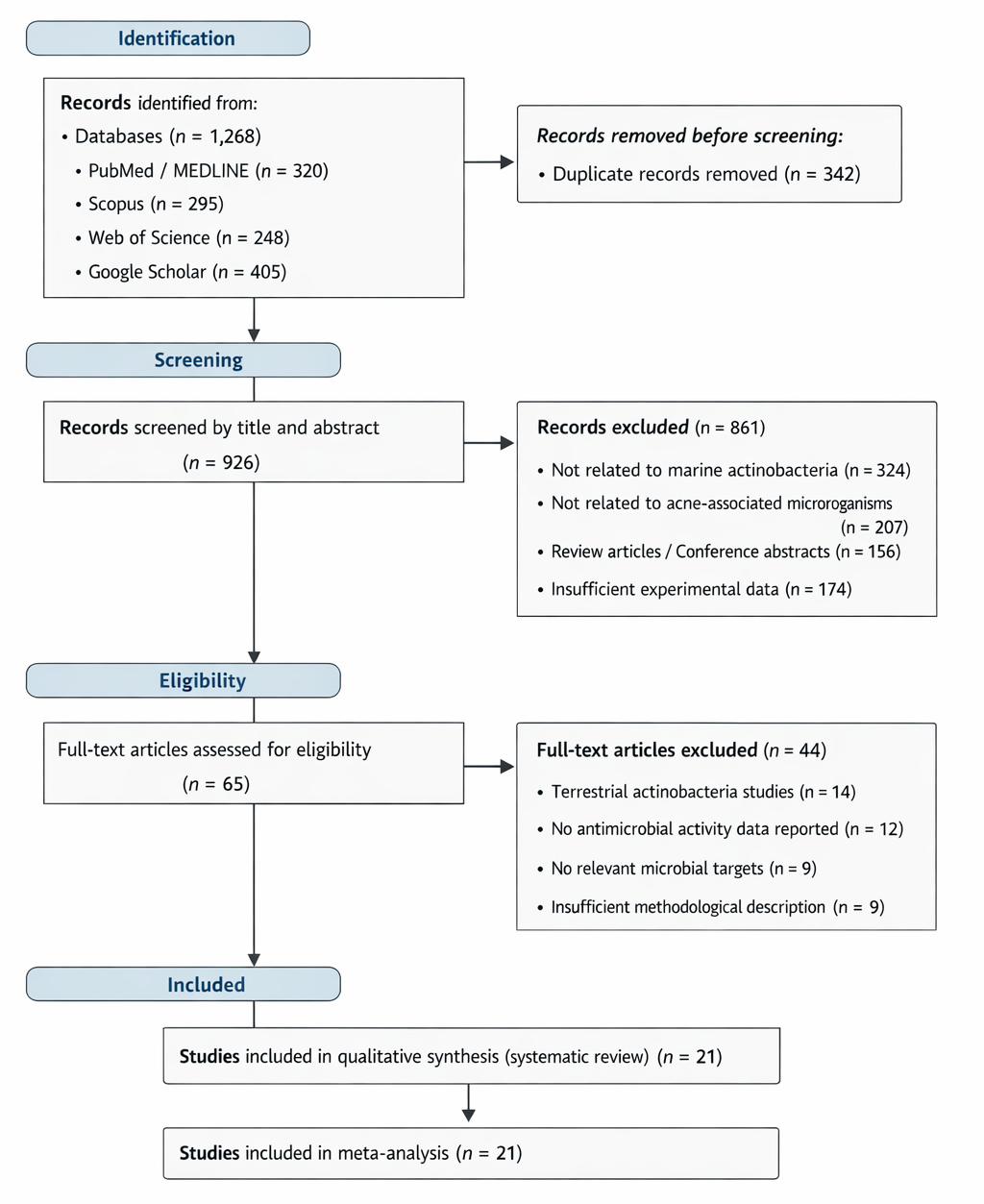
Systematic reviews provide a valuable framework for examining such emerging research landscapes. By synthesizing evidence across multiple studies, they can identify recurring patterns, highlight methodological limitations, and reveal areas where knowledge remains incomplete. Guidelines such as the PRISMA framework were developed precisely to improve the transparency and rigor of evidence synthesis in biomedical research (Moher et al., 2009). Applying such systematic approaches to the study of marine actinobacteria may help clarify their relevance for dermatological applications and guide future experimental work.
Against this backdrop, the present review seeks to explore marine actinobacteria as potential anti-infective resources for acne vulgaris. Rather than focusing solely on antibacterial potency, this perspective considers the broader ecological and microbiological context of acne pathogenesis, including microbial dysbiosis, biofilm formation, and antimicrobial resistance. By examining current evidence through a systematic review lens, the study aims to identify promising compounds, evaluate emerging therapeutic strategies, and highlight research gaps that may shape the next generation of acne treatments.