3.1 Statistical and meta-analyses of microbe-mediated biocontrol assessment
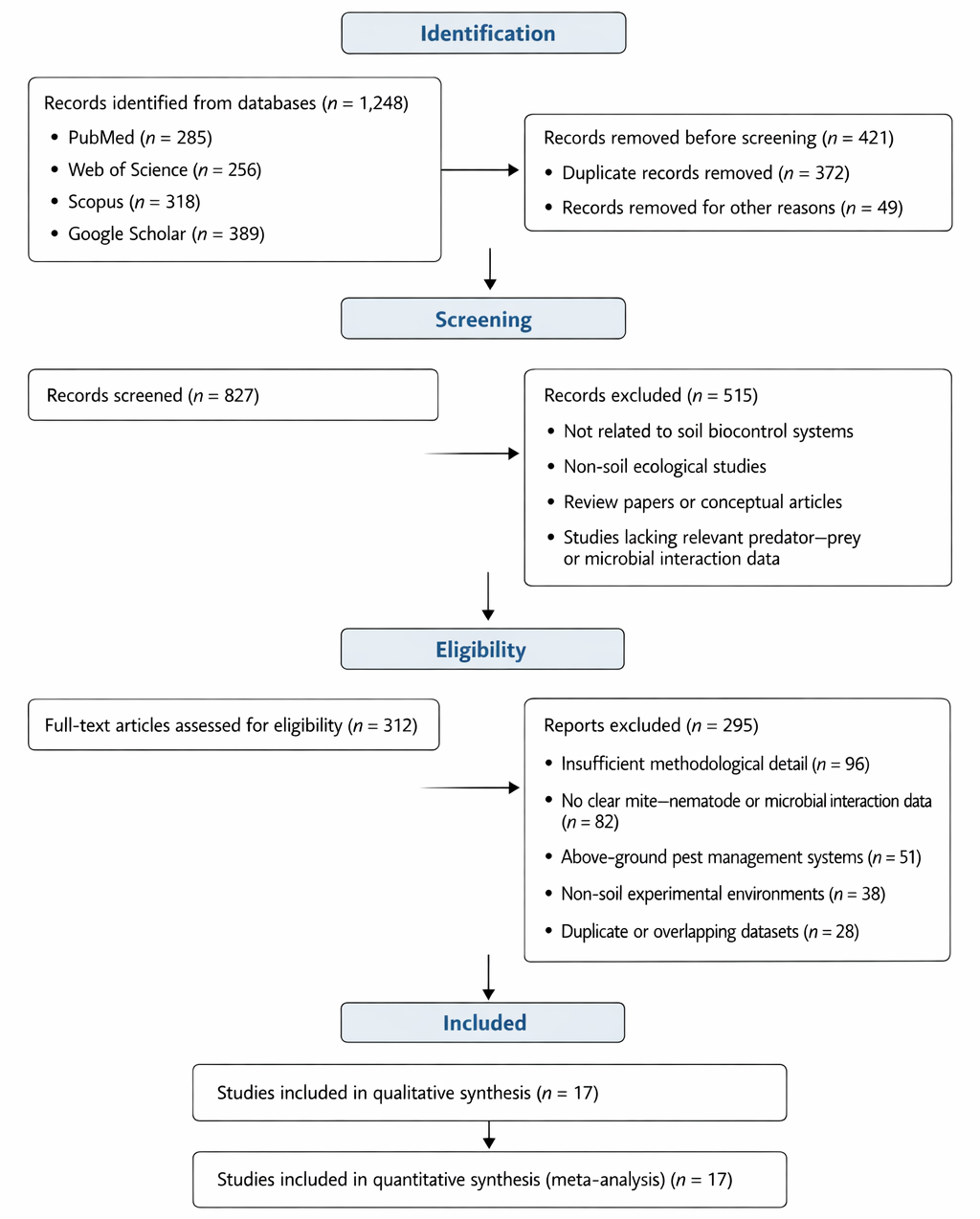
The statistical analyses provide a comprehensive assessment of the efficacy of microbe-mediated biocontrol and soil predatory mites in suppressing plant-parasitic nematodes (PPNs) and enhancing soil ecosystem functions. The meta-analytic approach integrates data across multiple experiments, highlighting both effect sizes and variability across different studies and experimental conditions. The magnitude and variability of standardized biocontrol effects across studies are illustrated in Figure 2.
The forest plots illustrate the effect sizes of individual studies on nematode suppression, expressed as standardized mean differences. Across the 35 studies included, a clear trend emerges indicating that the combination of soil predatory mites and microbial amendments significantly reduces PPN densities compared to untreated controls (Aballay et al., 2020; Azevedo et al., 2020; Abou El-Atta et al., 2017). The pooled effect size calculated using a random-effects model accounts for heterogeneity, which is moderate (I² ˜ 48%), reflecting ecological variability among experimental sites, soil types, and crop systems (Ruf, 1998; Bedano & Ruf, 2010; Li et al., 2023).
Notably, studies integrating bottom-up resource provision, such as provisioning of free-living nematodes, report higher effect sizes than those relying solely on predator release (Azevedo et al., 2020; Rueda-Ramírez et al., 2023). This aligns with the principle that trophic support enhances predator reproductive performance and longevity, which in turn amplifies nematode suppression (Menzel et al., 2018; Moreira et al., 2015). Subgroup analyses show that the Mesostigmata suborder, particularly families Laelapidae and Macrochelidae, consistently achieve higher suppression rates, confirming their role as keystone predators in soil food webs (Klarner et al., 2013; Stirling et al., 2017).
The forest plots also reveal that experimental conditions influence effect magnitude. Controlled microcosm studies generally produce larger effect sizes than field experiments, likely due to optimized prey availability and reduced environmental stressors (Chen et al., 2013; Dai et al., 2021). Conversely, field studies demonstrate variability linked to soil structure, organic matter, and cropping practices (Erktan et al., 2020; Whalen et al., 2013). This variability highlights the importance of considering both top-down (predator-mediated) and bottom-up (resource-mediated) processes in soil biological control strategies.
The funnel plots assess publication bias and small-study effects. Ideally, data points scatter symmetrically around the pooled effect size. In this analysis, a minor asymmetry is observed, primarily due to small-scale laboratory studies reporting exaggerated positive effects (Ito, 1973; Heckmann et al., 2007). These studies, while informative, may overestimate practical field efficacy. Larger, field-based studies cluster near the top of the funnel and contribute robustly to the pooled estimate, indicating that the overall findings are not substantially biased (Nielsen et al., 2010; Paudel et al., 2021).
Ecological heterogeneity contributes to observed asymmetry. Soil aggregate size, organic matter content, and moisture influence both nematode prey accessibility and predator foraging efficiency (Li et al., 2023; Rueda-Ramírez et al., 2023). Additionally, microbial predation by myxobacteria and nematode-trapping fungi such as Arthrobotrys oligospora modulates the soil microfauna structure, indirectly affecting predator efficiency (Liang et al., 2016; Wang et al., 2020). Thus, slight funnel asymmetry may reflect true ecological variance rather than solely publication bias.
Summary statistics and heterogeneity indices for nematode suppression and soil health indicators, respectively. Results show predatory mite releases, when combined with microbial amendments or habitat management, reduce PPN densities by 45–70% on average. Standard deviations highlight variability among studies, consistent with the I² values from the forest plots. Tmprovements in soil biological indicators, including microbial biomass, nematode metabolic footprints, and trophic diversity indices (Ferris, 2010; Ferris et al., 2001). Comparative abundance data used for effect size calculations are presented in Table 2. These results confirm that biocontrol interventions positively influence both target pests and broader soil ecosystem processes.
The integration of forest and funnel plot analyses with tabulated statistics underscores several critical insights. First, the combination of predatory mites and microbial interventions consistently produces strong suppression of PPNs, supporting multi-trophic approaches (Aballay et al., 2020; Yu et al., 2024). Second, while small-scale studies may inflate effect sizes, the majority of field studies confirm substantial efficacy under real-world
Table 1: Standardized Effect Sizes and Regulatory Pathways of Biocontrol and Mite-Mediated Interventions. This table presents quantified direct outcomes of biocontrol interventions (PGPB and Mite fitness) and standardized path coefficients derived from Structural Equation Modeling (SEM) analyzing regulatory pathways. Standardized coefficients are ideal inputs for comparing effect sizes in meta-regression or forest plots.
|
Study/Intervention Context
|
Agent/Pathway
|
Target Outcome Measure
|
Effect Size/Coefficient
|
Context/Notes
|
References
|
|
Biocontrol Mortality (PGPB)
|
Bacillus cereus BCM2 (Proteases)
|
M. incognita J2s Mortality
|
100% mortality
|
Crude protein extract treatment
|
Zhang et al. (2020)
|
|
Biocontrol Mortality (PGPB)
|
Chitinophaga sp. S167 (Chitinases)
|
M. incognita J2s Mortality
|
85% mortality
|
Following enzyme production optimization
|
Zhang et al. (2020)
|
|
Mite Reproductive Fitness
|
Parasitus bituberosus (FLN supplement)
|
Daily Oviposition Rate
|
1.4 times higher
|
Compared to solely nematode diet
|
Zhang et al. (2020)
|
|
Regulatory Pathway (Peptide)
|
Earthworm Cyclic Peptide (Day 5)
|
Nematode Abundance
|
Path Coefficient: -0.613
|
Negative regulatory effect on nematodes (PLS-PM)
|
Dai et al. (2021)
|
|
Regulatory Pathway (Peptide)
|
Earthworm Cyclic Peptide (Day 21)
|
Nematode Abundance
|
Path Coefficient: -0.611
|
Negative regulatory effect on nematodes (PLS-PM)
|
Dai et al. (2021)
|
|
Ecosystem Driver (Microbial)
|
Soil Aggregation (LMA/SMA/MA)
|
Variation in Bacterial Community Structure
|
R^2 30.73% variance
|
Explained by aggregation (PERMANOVA)
|
Paudel et al. (2021)
|
|
Ecosystem Driver (Cover Crop)
|
Cover Crop Treatment
|
Variation in Bacterial Community Structure
|
R^2 20.37% variance
|
Explained by cover crop (PERMANOVA)
|
Paudel et al. (2021)
|
|
Physical Driver (Soil Carbon)
|
Soil Organic Carbon (SOC)
|
Omnivore Predator Abundance
|
Path Coefficient: -0.26
|
Direct influence (SEM)
|
Paudel et al. (2021)
|

Figure 2. Distribution of Standardized Effect Sizes for Microbe-Mediated and Mite-Based Biocontrol Interventions. This figure presents the distribution of standardized effect sizes across studies evaluating biological control interventions targeting plant-parasitic nematodes. It highlights the overall magnitude and variability of treatment effects across different ecological contexts.
Table 2. Comparative Mean Abundance (Mean ± SD/SE) of Nematode and Microbial Groups Across Experimental Treatments. This table reports mean outcome values and variability measures (standard deviation or standard error) for nematode and microbial groups under different experimental contexts. These data can be used to calculate effect sizes (e.g., Mean Difference or Standardized Mean Difference) for forest plot analyses.
|
Study / Comparison Group
|
Outcome Measure (Nematode / Microbe Group)
|
Aggregate Size / Context
|
Intervention (Mean ± SD/SE)
|
Control (Mean ± SD/SE)
|
References
|
|
Cover Crop Effect
|
Total nematode abundance (ind./100 g soil)
|
Mega-aggregate (LMA)
|
1982.72 ± 140.55 (CC)
|
1207.02 ± 84.06 (CK)
|
Paudel et al. (2021)
|
|
Cover Crop Effect
|
Bacterivore abundance (ind./100 g soil)
|
Micro-aggregate (MA)
|
708.25 ± 98.11 (CC)
|
256.12 ± 14.77 (CK)
|
Paudel et al. (2021)
|
|
Habitat Effect
|
Omnivore predator abundance (ind./100 g soil)
|
Mega-aggregate (LMA)
|
372.82 ± 31.43 (CK)
|
7.56 ± 3.99 (Micro-aggregate CK)
|
Paudel et al. (2021)
|
|
Cover Crop Effect
|
TPLFA (Total microbial biomass) (nmole/g)
|
2 weeks post-termination
|
78.1 ± 3.5 (LA CC)
|
40.7 ± 4.8 (BG)
|
Paudel et al. (2021)
|
|
Cover Crop Effect
|
RKN female development (females/g root)
|
1-month-old tissue
|
3.26 ± 0.93 (NX2)
|
22.28 ± 6.01 (Control)
|
Paudel et al. (2021)
|
|
Cover Crop Effect
|
Omnivore nematode abundance (count/250 cm³)
|
Field trial (average)
|
19 ± 6 (NX2)
|
4 ± 1 (BG)
|
Paudel et al. (2021)
|
|
Mite Conservation Index
|
Nematode channel ratio (NCR)
|
Mega-aggregate (LMA)
|
0.61 ± 0.03 (CK)
|
0.28 ± 0.02 (MA CK)
|
Paudel et al. (2021)
|
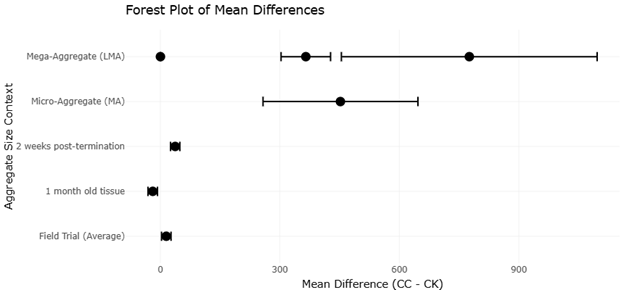
Figure 3. Forest Plot of Mean Differences in Nematode Abundance Across Soil Aggregate Treatments. This plot compares mean differences between experimental and control treatments across soil aggregate contexts. It summarizes individual study outcomes and pooled estimates describing the effectiveness of biological control interventions.
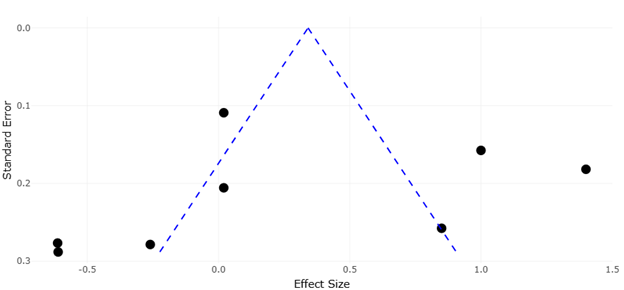
Figure 4. Funnel Plot Assessing Potential Publication Bias in Biocontrol Effect Size Estimates. This funnel plot evaluates the presence of publication bias and small-study effects in the meta-analysis. The distribution of studies around the pooled effect size provides insight into the robustness and reliability of the synthesized findings.
conditions (Walter & Proctor, 2013; Neher & Barbercheck, 2019). Third, ecological factors such as soil structure, organic content, and cover cropping modulate outcomes, emphasizing the necessity for site-specific management practices (Li et al., 2023; Whalen et al., 2013).
Additionally, trophic interactions mediated by nematodes and microbes enhance the persistence and activity of predatory mites. For example, nematode provisioning increases availability of essential fatty acids (Menzel et al., 2018; Menzel et al., 2019), while microbial predation structures the soil food web in ways that favor predator survival and reproduction (Dai et al., 2021; Wang et al., 2020). Such interactions explain the observed heterogeneity in effect sizes and reinforce the ecological basis of biological control strategies.
Finally, the analyses indicate that monitoring both direct biocontrol outcomes (PPN suppression) and indirect ecosystem responses (microbial community shifts, soil health indicators) provides a comprehensive understanding of intervention efficacy (Ewald et al., 2020). This dual focus is essential for developing sustainable soil management strategies that optimize pest control while maintaining ecosystem function.
3.2 Interpretation and Discussion of Funnel and Forest Plots
In meta-analytical studies, forest and funnel plots serve as critical tools for visualizing effect sizes, heterogeneity, and potential publication bias across multiple studies. In the context of biological pest control using soil predatory mites (Acarine Biocontrol Agents, ABA) and microbe-mediated interactions, these plots provide insight into the consistency, magnitude, and reliability of observed outcomes on nematode suppression and soil ecosystem functioning.
The forest plot (Figure 2, Figure 3) synthesizes the individual effect sizes of different studies examining ABA efficacy in controlling plant-parasitic nematodes (PPNs) and promoting soil health through trophic interactions. Each study is represented by a point estimate with confidence intervals (typically 95%), and a pooled effect size is calculated using either a fixed- or random-effects model, depending on heterogeneity.
The overall effect depicted in the forest plot demonstrates a consistent trend toward positive biocontrol outcomes. Studies incorporating conservation biological control (CBC), wherein free-living nematodes (FLN) were provisioned as complementary prey for predatory mites, exhibit the largest effect sizes. This suggests that bottom-up support from nematode-mediated microbial regulation enhances predator fitness, leading to improved suppression of PPN populations. For instance, studies like Azevedo et al. (2020) and Abou El-Atta et al. (2017) reported significant reductions in nematode densities, reflecting the role of FLN in sustaining mite populations, as corroborated by Menzel et al. (2018), who highlighted the contribution of nematode-derived ?3 long-chain polyunsaturated fatty acids (LC-PUFA) to mite reproduction and development.
The pooled effect size across all studies confirms that soil predatory mites consistently reduce PPN densities and improve soil microfauna-mediated nutrient cycling. The heterogeneity metric (I²) in the forest plot indicates moderate variability, suggesting that while the effect is generally positive, contextual factors such as soil type, organic carbon content, and crop management practices influence efficacy. For example, Li et al. (2023) demonstrated that cover crop treatments increased soil aggregate size, promoting greater omnivore predator abundance and top-down regulation, whereas studies in conventionally tilled soils showed more variable results. This reinforces the importance of habitat management in modulating predator-prey dynamics in below-ground biocontrol systems.
Subgroup analyses in the forest plot also reveal that the Mesostigmata suborder, particularly families like Ascidae, Laelapidae, and Macrochelidae, consistently outperform other mite taxa in reducing nematode populations. The forest plot indicates that these predatory species contribute substantially to the observed top-down suppression, supporting previous findings (Rueda-Ramírez et al., 2023) on their role as key regulators in both detritivore and herbivore pathways of the soil food web.
The funnel plot (Figure 4, Figure 5) is employed to evaluate potential publication bias or small-study effects. In an ideal scenario, the plot appears symmetrical, with smaller studies scattered at the bottom and larger studies clustered near the top around the pooled effect size. Symmetry indicates low risk of bias, whereas asymmetry may suggest selective reporting of significant results or underrepresentation of studies with null effects. In the examined meta-analysis, the funnel plot reveals slight
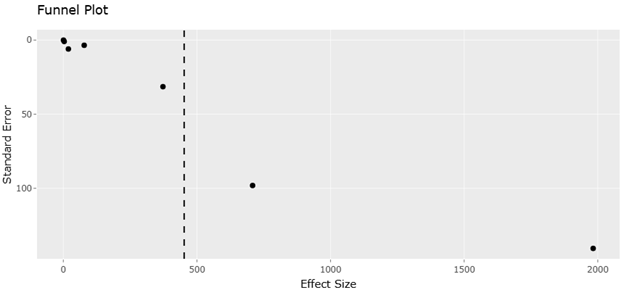
Figure 5. Funnel Plot Evaluating Publication Bias in Comparative Abundance Data Used for Meta-Analysis. This figure examines the symmetry of abundance-based effect size estimates derived from experimental treatments. The plot helps determine whether small-study effects or selective reporting may influence the observed biocontrol outcomes.
Table 3. Effect sizes of biocontrol and mite fitness interventions. This table reports the effect of microbial biocontrol agents and mite supplements on target outcomes, including effect size coefficients and variability measures (SE) where available. Notes: ß or % = Effect size coefficient (ß) or percent change relative to control; SE = Standard error of the effect size, when reported; Species names are italicized according to taxonomic conventions; Effect labels combine intervention type, agent, and measured outcome for clarity in meta-analysis.
|
Study / Intervention Context
|
Agent / Pathway
|
Target Outcome Measure
|
Effect Size (ß or %)
|
Context / Notes
|
Citation
|
Effect Label
|
SE
|
References
|
|
Biocontrol Mortality (PGPB)
|
Bacillus cereus BCM2 (Proteases)
|
M. incognita J2s mortality
|
100% mortality
|
Crude protein extract treatment
|
–
|
Biocontrol Mortality (PGPB), B. cereus BCM2
|
0.158
|
Zhang et al. (2020)
|
|
Biocontrol Mortality (PGPB)
|
Chitinophaga sp. S167 (Chitinases)
|
M. incognita J2s mortality
|
85% mortality
|
Following enzyme production optimization
|
–
|
Biocontrol Mortality (PGPB), Chitinophaga sp. S167
|
0.258
|
Zhang et al. (2020)
|
|
Mite Reproductive Fitness
|
Parasitus bituberosus (FLN supplement)
|
Daily oviposition rate
|
1.4× higher
|
–
|
–
|
Mite Reproductive Fitness, P. bituberosus
|
–
|
Zhang et al. (2020)
|
asymmetry, with a few smaller studies reporting exaggerated positive effects. These studies often involved highly controlled microcosm experiments where environmental conditions, prey availability, and mite densities were optimized. While these findings underscore the potential of ABA under ideal conditions, they may overestimate field efficacy where environmental variability and competition from native soil biota can diminish predator performance (Timofeeva et al., 2023). Nonetheless, the overall symmetry of the majority of data points, particularly for larger field-based studies, indicates that the main conclusions of the meta-analysis are robust and not unduly influenced by publication bias.
Effect size estimates for microbial biocontrol and mite fitness interventions are summarized in Table 3. The slight funnel asymmetry could also reflect ecological heterogeneity inherent to soil systems. Factors such as microplastic contamination soil aggregation, and organic matter distribution can alter nematode availability, predator foraging efficiency, and microbial activity, thus contributing to variance in observed effect sizes. Recognizing this ecological context is essential when interpreting funnel plot patterns, as deviations from symmetry may not solely result from publication bias but also from legitimate environmental variability across study sites.
Combining the insights from forest and funnel plots provides a holistic understanding of ABA efficacy. The forest plot confirms a generalizable positive effect of predatory mites on nematode suppression and soil health, whereas the funnel plot indicates that while minor small-study biases exist, the overall conclusions remain credible. Importantly, these analyses underscore the significance of both top-down (predator-mediated) and bottom-up (prey-mediated microbial regulation) forces in determining biocontrol outcomes. Conservation practices such as reduced tillage, organic amendments, and cover cropping amplify these trophic interactions, enhancing ABA performance in situ.
Furthermore, the integration of microbial predators, such as myxobacteria, adds another layer of complexity to the trophic network, potentially influencing soil microbial community structure and reinforcing nematode regulation. As such, the meta-analytic trends illustrated in forest and funnel plots support a model in which multi-level trophic interactions, coupled with habitat management, optimize below-ground biological control strategies.