Afridi, M. S., Ali, S., Salam, A., César Terra, W., Hafeez, A., Sumaira, Ali, B., AlTami, M. S., Ameen, F., Ercisli, S., Marc, R. A., Medeiros, F. H. V., & Karunakaran, R. (2022). Plant microbiome engineering: Hopes or hypes. Biology, 11(12), 1782. https://doi.org/10.3390/biology11121782
Calatrava, V., Tejada-Jimenez, M., Sanz-Luque, E., Fernandez, E., Galvan, A., & Llamas, A. (2023). Chlamydomonas reinhardtii, a reference organism to study algal–microbial interactions: Why can’t they be friends? Plants, 12(4), 788. https://doi.org/10.3390/plants12040788
Croft, M. T., Lawrence, A. D., Raux-Deery, E., Warren, M. J., & Smith, A. G. (2005). Algae acquire vitamin B12 through a symbiotic relationship with bacteria. Nature, 438(7064), 90–93. https://doi.org/10.1038/nature04056
Enamala, M. K., Enamala, S., Chavali, M., Donepudi, J., Yadavalli, R., Kolapalli, B., Aradhyula, T. V., Velpuri, J., & Kuppam, C. (2018). Production of biofuels from microalgae: A review on cultivation, harvesting, lipid extraction, and numerous applications of microalgae. Renewable and Sustainable Energy Reviews, 94, 49–68. https://doi.org/10.1016/j.rser.2018.05.012
Enamala, M. K., Enamala, S., Chavali, M., Donepudi, J., Yadavalli, R., Kolapalli, B., Aradhyula, T. V., Velpuri, J., & Kuppam, C. (2018). Production of biofuels from microalgae - A review on cultivation, harvesting, lipid extraction, and numerous applications of microalgae. Renewable and Sustainable Energy Reviews, 94, 49–68. https://doi.org/10.1016/j.rser.2018.05.059
Habib, A., Nishi, S., Haque, M. M., Tauhiduzzaman, M., Khatun, K., Akter, M. S., & Ghosh, G. C. (2025b). Adsorption and desorption of methyl orange dye on environmentally aged polyethylene, polyethylene terephthalate and polystyrene microplastics in aquatic environment. Plos one, 20(7), e0323516. https://doi.org/10.1371/journal.pone.0323516
Habib, A., Rahman, M. A., Akter, M. S., Chakraborty, T. K., Zaman, S., & Ghosh, G. C. (2025a). Microplastics in Beach Sediments of the Northern Bay of Bengal, Bangladesh: Insights into Occurrence, Distribution, Pollution Indices, and ANN-Based Risk Modeling. Journal of Hazardous Materials: Plastics, 100008. https://doi.org/10.1016/j.hazmp.2025.100008
Ling, J., Nip, S., Cheok, W. L., de Toledo, R. A., & Shim, H. (2014). Lipid production by a mixed culture of oleaginous yeast and microalga from distillery and domestic mixed wastewater. Bioresource Technology, 173, 132–139. https://doi.org/10.1016/j.biortech.2014.09.047
Lucia, U., & Grisolia, G. (2021). Biofuels from micro-organisms: Thermodynamic considerations on the role of electrochemical potential on micro-organisms growth. Applied Sciences, 11(6), 2591. https://doi.org/10.3390/app11062591
Makut, B. B., Goswami, G., & Das, D. (2020). Evaluation of bio-crude oil through hydrothermal liquefaction of microalgae-bacteria consortium grown in open pond using wastewater. Biomass Conversion and Biorefinery. https://doi.org/10.1007/s13399-020-00705-y
Mettler, T., Mühlhaus, T., Hemme, D., Schöttler, M. A., Rupprecht, J., Idoine, A., Veyel, D., Pal, S. K., Yaneva-Roder, L., Winck, F. V., et al. (2014). Systems analysis of the response of photosynthesis, metabolism, and growth to an increase in irradiance in the photosynthetic model organism Chlamydomonas reinhardtii. Plant Cell, 26, 3446–3467. https://doi.org/10.1105/tpc.114.124537
Musa, M., Ayoko, G. A., Ward, A., Rösch, C., Brown, R. J., & Rainey, T. J. (2019). Factors affecting microalgae production for biofuels and the potentials of chemometric methods in assessing and optimizing productivity. Cells, 8, 815. https://doi.org/10.3390/cells8080851
Padmaperuma, G., Kapoore, R. V., Gilmour, D. J., & Vaidyanathan, S. (2018). Microbial consortia: A critical look at microalgae co-cultures for enhanced biomanufacturing. Critical Reviews in Biotechnology, 35, 690–703. https://doi.org/10.1080/07388551.2017.1390728
Papone, T., Kookkhunthod, S., & Leesing, R. (2012). Microbial oil production by monoculture and mixed cultures of microalgae and oleaginous yeasts using sugarcane juice as substrate. World Acad Sci Eng Technol, 64, 1127-1131.
Polle, J. E. W., Neofotis, P., Huang, A., Chang, W., Sury, K., & Wiech, E. M. (2014). Carbon partitioning in green algae (Chlorophyta) and the enolase enzyme. Metabolites, 4(3), 612–628. https://doi.org/10.3390/metabo4030612
Ramanan, R., Kim, B., Cho, D., & Kim, H. (2016). Algae-bacteria interactions: Evolution, ecology and emerging applications. Biotechnology Advances, 34, 14–29. https://doi.org/10.1016/j.biotechadv.2015.12.003
Ruan, K., Duan, J. B., Bai, F., Lemaire, M., Ma, X., & Bai, L. (2009). Function of Dunaliella salina (Dunaliellaceae) enolase and its expression during stress. European Journal of Phycology, 44, 207–214. https://doi.org/10.1080/09670260802573105
Scognamiglio, V., Giardi, M. T., Zappi, D., Touloupakis, E., & Antonacci, A. (2021). Photoautotrophs–bacteria co-cultures: Advances, challenges and applications. Materials, 14(11), 3027. https://doi.org/10.3390/ma14113027
Seymour, J. R., Amin, S. A., Raina, J. B., & Stocker, R. (2017). Zooming in on the phycosphere: The ecological interface for phytoplankton–bacteria relationships. Nature Microbiology, 2(10), 17065. https://doi.org/10.1038/nmicrobiol.2017.65
Shen, S., Wang, G., Zhang, M., Tang, Y., Gu, Y., Jiang, W., Wang, Y., & Zhuang, Y. (2020). Effect of temperature and surfactant on biomass growth and higher-alcohol production during syngas fermentation by Clostridium carboxidivorans P7. Bioresources and Bioprocessing, 7, 56. https://doi.org/10.1186/s40643-020-00344-4
Tan, L. T. (2023). Impact of marine chemical ecology research on the discovery and development of new pharmaceuticals. Marine Drugs, 21(3), 174. https://doi.org/10.3390/md21030174
Valenzuela Ruiz, V., Cervantes Enriquez, E. P., Vázquez Ramírez, M. F., Bivian Hernández, M. d. l. Á., Cárdenas-Manríquez, M., Parra Cota, F. I., & de los Santos Villalobos, S. (2025). A new era in the discovery of biological control bacteria: Omics-driven bioprospecting. Soil Systems, 9(4), 108. https://doi.org/10.3390/soilsystems9040108
Wei, Z., Wang, H., Li, X., Zhao, Q., Yin, Y., Xi, L., Ge, B., & Qin, S. (2020). Enhanced biomass and lipid production by co-cultivation of Chlorella vulgaris with Mesorhizobium sangaii under nitrogen limitation. Journal of Applied Phycology, 32, 233–242. https://doi.org/10.1007/s10811-019-01924-4
Xue, F., Miao, J., Zhang, X., & Tan, T. (2010). A new strategy for lipid production by mix cultivation of Spirulina platensis and Rhodotorula glutinis. Applied Biochemistry and Biotechnology, 160, 498–503. https://doi.org/10.1007/s12010-008-8376-z
Yao, S., Liu, S., An, Y., Lu, J., Gjermansen, C., & Schramm, A. (2018). Microalgae-bacteria symbiosis in microalgal growth and biofuel production: A review. Journal of Applied Microbiology, 126, 359–368. https://doi.org/10.1111/jam.14095
Zhang, S., Liu, Y., & Bryant, D. A. (2015). Metabolic engineering of Synechococcus sp. PCC 7002 to produce poly-3-hydroxybutyrate and poly-3-hydroxybutyrate-co-4-hydroxybutyrate. Metabolic Engineering, 32, 174–183. https://doi.org/10.1016/j.ymben.2015.09.012
Zhao, P., Yu, X., Li, J., Tang, X., & Huang, Z. (2014). Enhancing lipid productivity by co-cultivation of Chlorella sp. U4341 and Monoraphidium sp. FXY-10. Journal of Bioscience and Bioengineering, 118(1), 72–77. https://doi.org/10.1016/j.jbiosc.2013.12.014

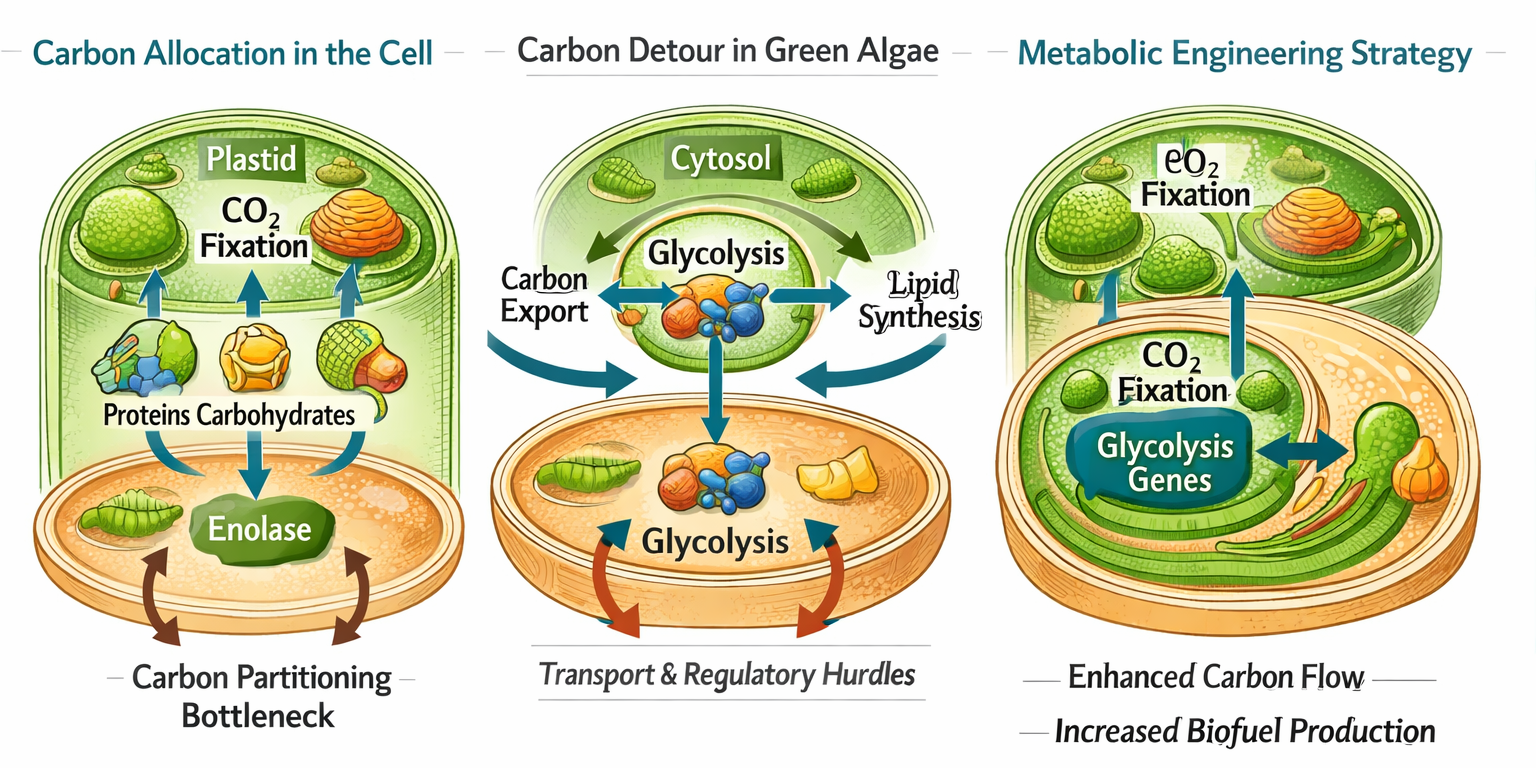
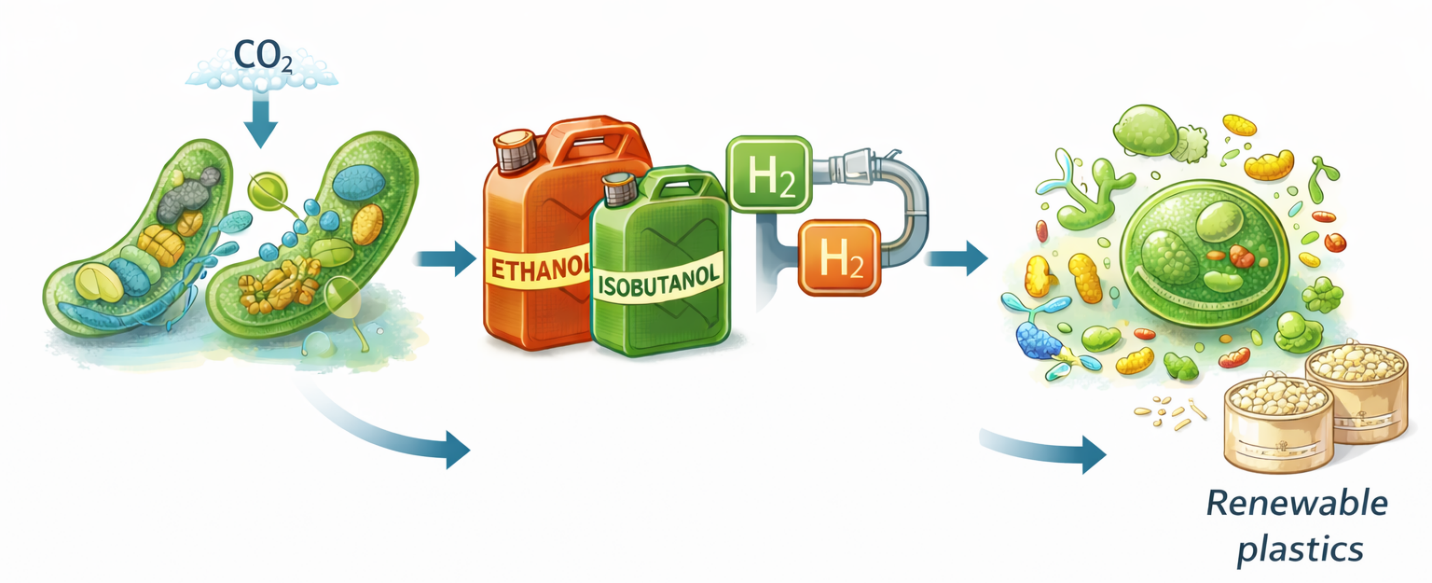
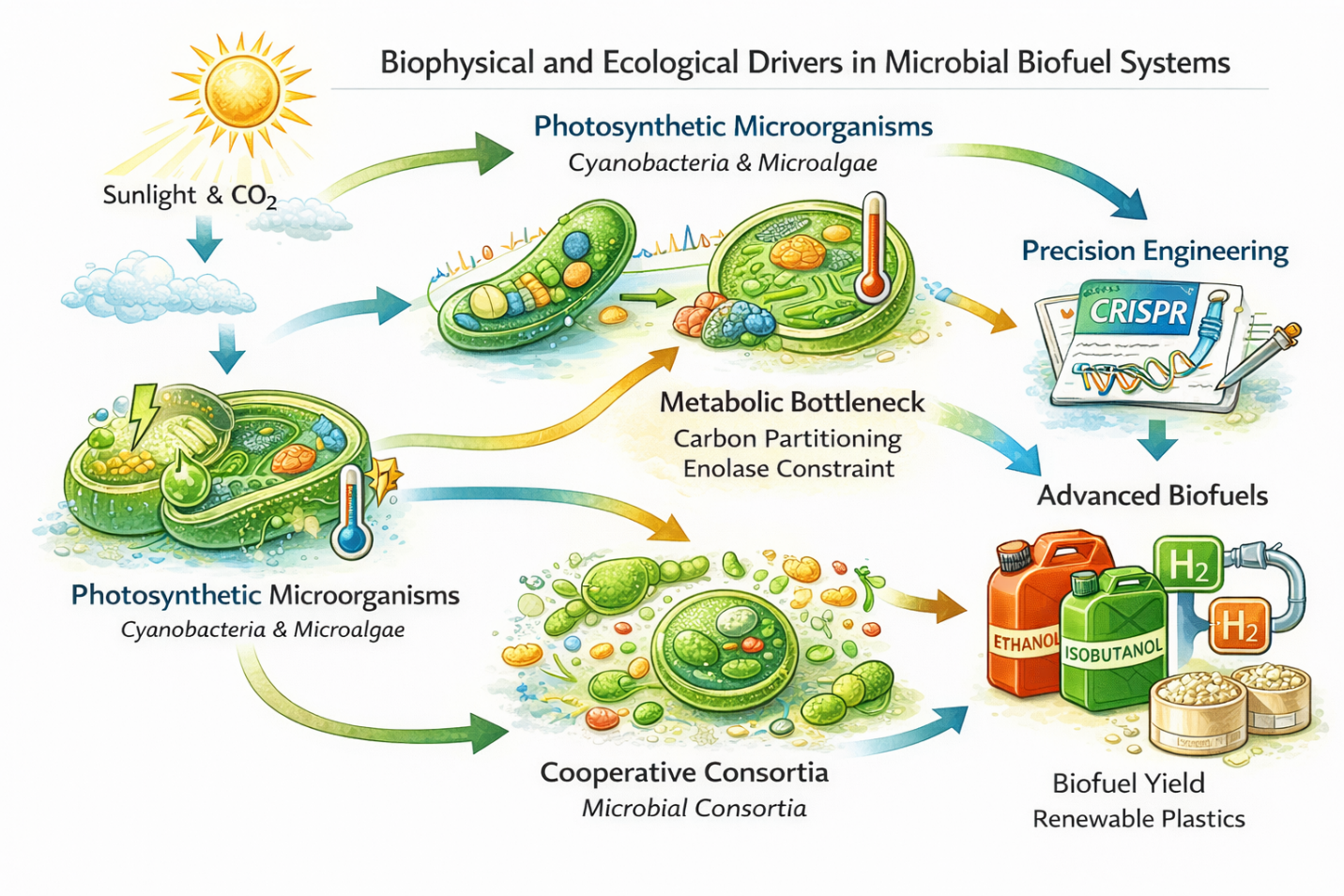 Figure 3: Integrated Biophysical and Ecological Drivers in Microbial Biofuel Production. Microbial biofuel productivity is shaped by the interaction of cellular energetics, metabolic carbon partitioning, microbial community interactions, and synthetic biology. Integrating thermodynamic principles, metabolic engineering, and cooperative consortia offers a pathway toward more efficient and sustainable biofuel systems.
Figure 3: Integrated Biophysical and Ecological Drivers in Microbial Biofuel Production. Microbial biofuel productivity is shaped by the interaction of cellular energetics, metabolic carbon partitioning, microbial community interactions, and synthetic biology. Integrating thermodynamic principles, metabolic engineering, and cooperative consortia offers a pathway toward more efficient and sustainable biofuel systems.