1. Introduction
Maternal mortality in the United States presents a paradox that is, in many ways, difficult to reconcile. Despite advances in clinical care, technology, and health system infrastructure, maternal deaths remain higher than in most other high-income nations. Even more concerning—perhaps more troubling—is the persistence of stark disparities. Black women, in particular, continue to experience maternal mortality rates that far exceed those of their White counterparts, even after adjusting for socioeconomic status and education (MacDorman et al., 2021). This suggests that the issue is not simply clinical, nor purely behavioral, but deeply embedded in structural and systemic conditions.
One of those conditions—often discussed, though perhaps still underestimated—is access. Access not only to prenatal care or specialist consultation, but to fully functional, hospital-based obstetric services. Over the past decade, rural hospital closures and the loss of obstetric units have quietly reshaped the maternal care landscape in the United States. In counties where obstetric services have disappeared, studies have shown worsening birth outcomes and reduced preparedness for emergencies (Kozhimannil et al., 2018). It is not difficult to imagine how, in such settings, even a relatively manageable complication could escalate rapidly.
And that brings us to a somewhat uncomfortable realization: maternal safety is not solely determined by clinical decision-making. It is also, quite critically, dependent on operational readiness. In obstetric emergencies—postpartum hemorrhage being a clear example—time is unforgiving. Survival depends on immediate access to blood products, uterotonic medications, skilled personnel, and functioning equipment. A delay of even minutes can change outcomes dramatically. Yet hospitals do not always operate under ideal conditions. Supply chain disruptions, staffing shortages, and procurement delays are not rare anomalies; they are, increasingly, part of the healthcare environment. National blood shortages, for instance, have exposed vulnerabilities that extend far beyond individual institutions (Saillant et al., 2022).
Interestingly, much of the maternal safety literature has not fully engaged with this operational dimension. The dominant focus has been on improving clinical protocols, enhancing quality improvement initiatives, and refining patient-level risk assessment. These are, without question, essential efforts. However, they may not be sufficient. A hospital may identify a patient as high-risk for hemorrhage—but if blood products are unavailable or staff are stretched beyond capacity, that knowledge alone does not translate into safety. In a sense, the system “knows,” but cannot act effectively.
This gap becomes even more evident when we consider the rapid rise of machine learning in obstetrics. Over the past several years, predictive models—particularly those based on gradient boosting and deep learning—have demonstrated impressive performance in forecasting adverse maternal outcomes. For example, models trained on electronic health record (EHR) data have shown strong ability to predict postpartum hemorrhage at the time of admission (Venkatesh et al., 2020). Similarly, large-scale deep learning frameworks have illustrated how predictive systems can be deployed across multiple institutions with high accuracy (Rajkomar et al., 2018).
And yet, despite these advances, there is something missing. Most of these models stop at prediction. They produce a risk score—a probability, a classification—but do not extend into the operational domain. Hospitals, however, do not respond to probabilities in isolation. They respond to demand. A predicted increase in hemorrhage risk should, ideally, trigger adjustments in blood inventory, staffing allocation, and supply chain coordination. But such integration is rarely implemented in practice. The result is a disconnect between what the system predicts and what it can operationally deliver.
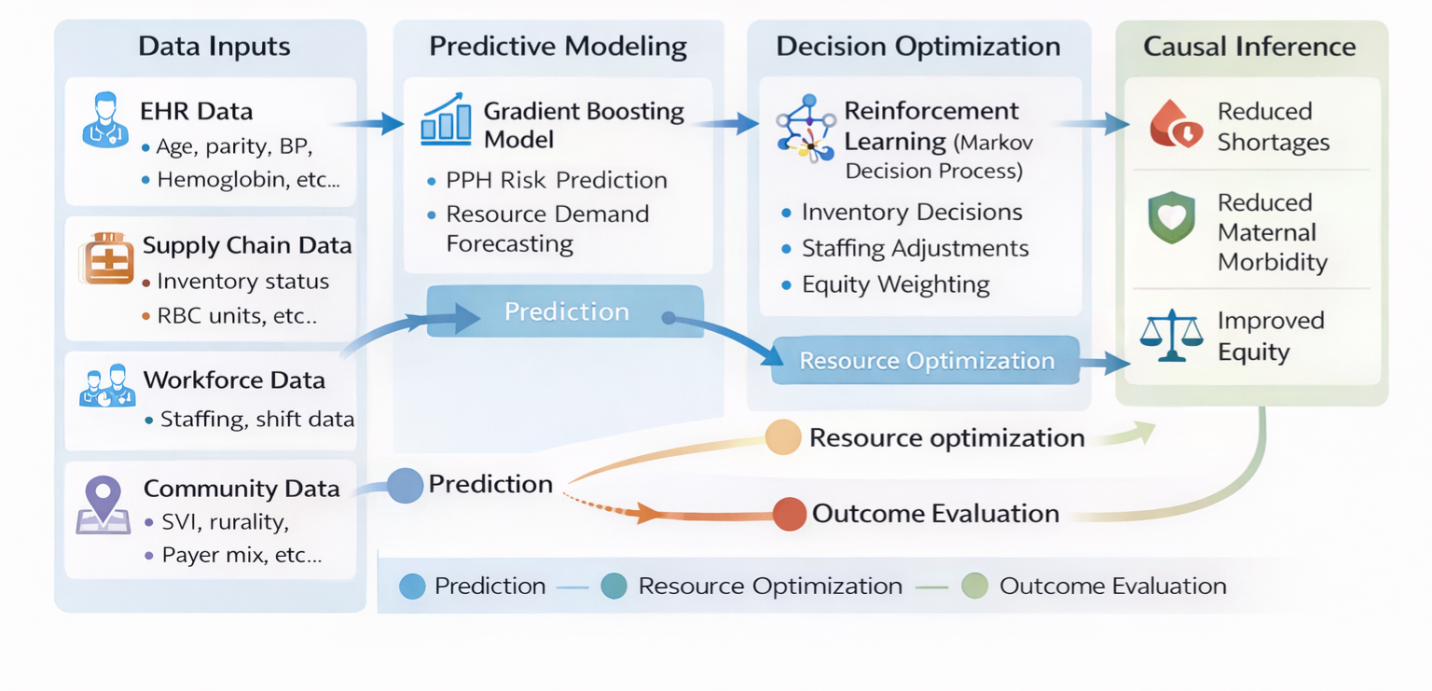
Addressing this disconnect requires a shift in perspective—from prediction to decision intelligence. Healthcare operations, particularly in high-stakes environments like obstetrics, are inherently dynamic. Resource allocation decisions must be made sequentially, under uncertainty, and often with incomplete information. This is precisely the kind of problem that reinforcement learning (RL) is designed to address. By framing resource management as a sequential decision-making process, RL allows systems to adapt to changing conditions, learning policies that optimize outcomes over time (Gottesman et al., 2019).
Still, predictive accuracy and optimization alone do not fully resolve the issue. There is a deeper methodological challenge—one that relates to causation. Better-resourced hospitals may naturally experience fewer adverse outcomes, but this does not necessarily mean that resource availability alone causes those outcomes. Confounding factors—such as baseline infrastructure, staffing levels, or patient population characteristics—can complicate interpretation. Without careful analysis, predictive systems risk reinforcing existing inequities rather than addressing them.
This is where causal inference becomes essential. Marginal structural models (MSMs), for example, provide a framework for estimating causal effects in the presence of time-varying confounding (Robins et al., 2000). By incorporating such methods, it becomes possible to move beyond correlation and begin to understand whether—and to what extent—resource adequacy directly influences maternal outcomes.
At the same time, any meaningful solution must also account for equity. Structural vulnerability is not evenly distributed across populations or geographies. Communities with higher levels of socioeconomic disadvantage, as captured by indices such as the Social Vulnerability Index (SVI), often face compounded risks—limited access, constrained resources, and heightened exposure to systemic barriers (Flanagan et al., 2011). If predictive and optimization systems do not explicitly incorporate these factors, they risk perpetuating, or even amplifying, disparities.
Taken together, these considerations point toward the need for a more integrated approach—one that connects clinical prediction, operational decision-making, causal evaluation, and equity prioritization. This study, therefore, proposes a closed-loop maternal resource intelligence framework. Rather than treating prediction, resource allocation, and outcome evaluation as separate processes, the framework links them into a continuous cycle. Predictive models estimate demand; reinforcement learning optimizes resource allocation in real time; and causal analysis evaluates the impact of those decisions on maternal outcomes.
The goal is not merely to improve predictive accuracy, but to enhance system responsiveness. To move, perhaps, from a reactive model of care—where hospitals respond to crises as they occur—to a more anticipatory, resilient system. One that recognizes that maternal safety is shaped not only by what clinicians know, but by what healthcare systems are prepared to do.
In a broader sense, this work aligns with national efforts to address maternal health disparities and improve quality of care. However, it also suggests that achieving these goals may require expanding our definition of quality itself. Clinical excellence, while essential, must be complemented by operational robustness. Without it, even the most advanced predictive tools may fall short of their potential.