1. Introduction
Environmental degradation has increasingly become one of the defining challenges of the modern era. Rapid urban expansion, industrial intensification, and large-scale agricultural practices have collectively altered natural ecosystems in ways that are difficult to reverse. These anthropogenic pressures have disrupted ecological stability, reduced habitat connectivity, and accelerated biodiversity loss across terrestrial and aquatic systems (Hanski, 1999). At the same time, climatic shifts associated with global warming are further amplifying environmental stressors, creating complex interactions between ecological disturbances and anthropogenic activities (Quereda Sala et al., 2000). Taken together, these pressures have intensified the search for sustainable technologies capable of mitigating environmental damage while maintaining economic productivity.
One particularly concerning issue involves the widespread contamination of natural environments by emerging pollutants. Pharmaceuticals and personal care products (PPCPs), for instance, have been detected in aquatic ecosystems worldwide, often at trace concentrations yet with potentially significant ecological consequences (Patel et al., 2019). Conventional wastewater treatment processes frequently fail to completely remove these compounds, allowing them to accumulate in rivers, soils, and sediments (Wang & Wang, 2016). This persistence raises questions about long-term ecological effects and underscores the need for environmentally friendly remediation strategies capable of degrading or transforming such contaminants.
In response to these challenges, global research and industrial communities have increasingly turned toward sustainability-oriented frameworks. Among these, Circular Supply Chain Management (CSCM) has emerged as a promising paradigm that promotes resource efficiency, waste minimization, and regenerative production systems (Farooque et al., 2019). Rather than treating industrial by-products as waste streams, circular approaches emphasize their transformation into valuable resources within closed-loop production systems. Such strategies are especially relevant in biotechnology, where organic residues can be converted into high-value biomolecules through microbial processes.
Parallel to these developments, advances in green technologies are reshaping the landscape of sustainable agriculture and environmental management. Nanotechnology, for example, has introduced innovative tools such as nano-biofertilizers that improve nutrient delivery and enhance crop resilience while reducing the ecological footprint of conventional fertilizers (Akhtar et al., 2022). These nanoscale materials possess high surface area and enhanced solubility, enabling more efficient nutrient uptake by plants and minimizing nutrient runoff into surrounding ecosystems (Nasrollahzadeh et al., 2019). Such approaches align closely with precision agriculture strategies aimed at optimizing productivity while preserving environmental integrity (Duhan et al., 2017). Within the broader context of sustainable agricultural policies, these technologies are increasingly being integrated into green development frameworks designed to reduce chemical inputs and environmental risks (Ginter, 2022).
Beyond agricultural applications, biotechnology offers powerful tools for transforming biomass into useful products. Microbial enzymes play a central role in this transformation, acting as biocatalysts capable of replacing energy-intensive or hazardous chemical processes. Enzymes such as cellulases and related lignocellulolytic enzymes facilitate the degradation of complex plant biomass into fermentable sugars, thereby supporting diverse industrial applications ranging from biofuel production to waste management (Kuhad et al., 2011). Their versatility has made them increasingly important across multiple sectors, including pharmaceuticals, food processing, and environmental remediation (Ejaz et al., 2021). The economic significance of industrial enzymes continues to grow as industries seek sustainable alternatives to conventional chemical treatments (Guerrand, 2018).
Among these enzymes, fungal laccases have attracted considerable attention due to their broad substrate specificity and ability to catalyze oxidation reactions involving phenolic and non-phenolic compounds. These multicopper oxidases are naturally produced by a variety of fungi, particularly white-rot species, and play an essential role in lignin degradation within forest ecosystems. Because of their oxidative capacity, laccases have been explored for numerous industrial applications including bioremediation, pulp and paper processing, textile treatment, and biosensor development. The challenge, however, lies in producing these enzymes efficiently and sustainably at industrial scale.
Solid-State Fermentation (SSF) has emerged as one of the most promising strategies for achieving this goal. Unlike submerged fermentation, SSF involves the cultivation of microorganisms on moist solid substrates with little or no free water, thereby closely mimicking the natural habitats of many fungi. This cultivation mode often leads to higher enzyme yields, improved stability, and lower energy requirements compared with liquid fermentation systems (Rehman et al., 2014). Moreover, SSF enables the direct utilization of agricultural residues as both physical support and nutrient source for microbial growth.
The use of agro-industrial waste materials as substrates represents a particularly attractive feature of SSF. Large quantities of lignocellulosic residues are generated globally from agricultural and food-processing industries, including rice straw, wheat straw, corn stover, sugarcane bagasse, and fruit processing by-products. Traditionally, these materials have been underutilized or disposed of through environmentally harmful practices such as open burning. However, their rich composition of cellulose, hemicellulose, and lignin makes them excellent substrates for microbial fermentation processes (Mussatto et al., 2012). Recent research has increasingly focused on converting these residues into valuable biomolecules, thereby contributing to sustainable waste management strategies (Šelo et al., 2021).
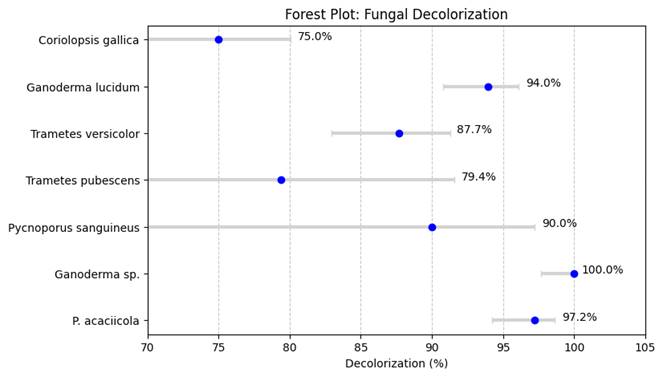
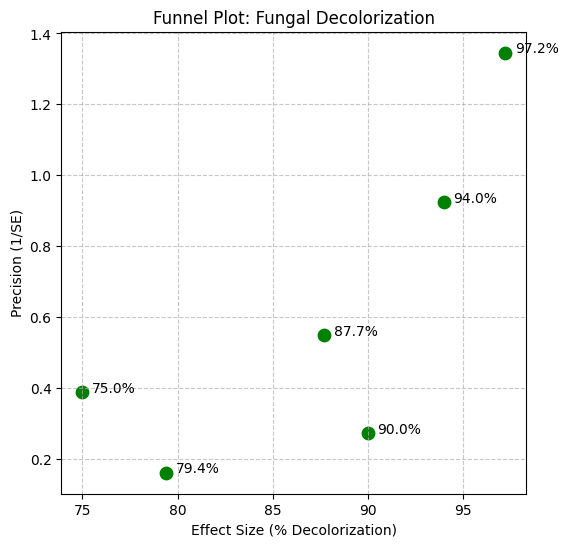
In the context of fungal fermentation, lignocellulosic substrates provide not only structural support but also act as natural inducers for enzyme production. Lignin and other aromatic compounds present in plant biomass can stimulate laccase synthesis by white-rot fungi, which utilize these enzymes to break down complex lignin polymers. Several studies have demonstrated that agricultural residues such as sugarcane bagasse, rice husk, and sunflower residues can effectively support fungal growth and enhance laccase production during SSF processes (Karp et al., 2015). Similarly, pilot-scale investigations have shown that rice straw and sunflower residues can be successfully converted into both medicinal mushrooms and laccase enzymes through controlled fermentation processes (Postemsky et al., 2017). In addition, the cultivation of edible and medicinal mushrooms on agro-industrial waste has been widely reported as an efficient strategy for producing lignocellulolytic enzymes while simultaneously generating valuable fungal biomass (Kumla et al., 2020).
In addition to enzyme production, the bioconversion of agro-industrial residues can generate a wide range of other valuable biomaterials. For example, lignocellulosic biomass can serve as a feedstock for biopolymers that are increasingly used in textile, packaging, and biomedical industries (Dias et al., 2025). Natural fibers derived from plant biomass are also gaining attention as sustainable alternatives to synthetic materials, particularly in industrial applications requiring biodegradable or renewable resources (Manaia et al., 2019). At the molecular level, microbial and plant-derived polymers are being explored for applications in advanced materials and biotechnology, further demonstrating the versatility of biomass-derived products (Rudin & Phillip, 2013).
Recent technological advances have expanded these possibilities even further. Electrospinning technologies, for instance, enable the fabrication of nanofibrous biomaterials capable of delivering therapeutic compounds in biomedical applications (Bonakdar & Rodrigue, 2024). Such nanofiber systems are increasingly investigated for controlled drug delivery and tissue engineering purposes (Luraghi et al., 2021). Similarly, bioactive compounds recovered from agricultural or winery by-products are being explored for their antimicrobial and antioxidant properties (Teixeira et al., 2014). Essential oils and plant-derived compounds have demonstrated promising antibacterial and antibiofilm activities, suggesting potential applications in food safety and medical biotechnology (Firmino et al., 2018).
Another emerging area involves microbial exopolysaccharides, which are extracellular polymers produced by bacteria and fungi. These biomolecules exhibit diverse functional properties, including emulsifying, antioxidant, and immunomodulatory activities, making them valuable in biomedical and food applications (Kaur & Dey, 2023). Advances in microbial biotechnology have revealed numerous exopolysaccharide-producing microorganisms capable of generating these materials under controlled fermentation conditions (Netrusov et al., 2023). In parallel, advances in biomaterial engineering and encapsulation technologies have enabled the controlled release of bioactive compounds from packaging and polymer matrices, highlighting new applications of fermentation-derived biomolecules in food preservation and biomedical systems (Siddiqui et al., 2023).
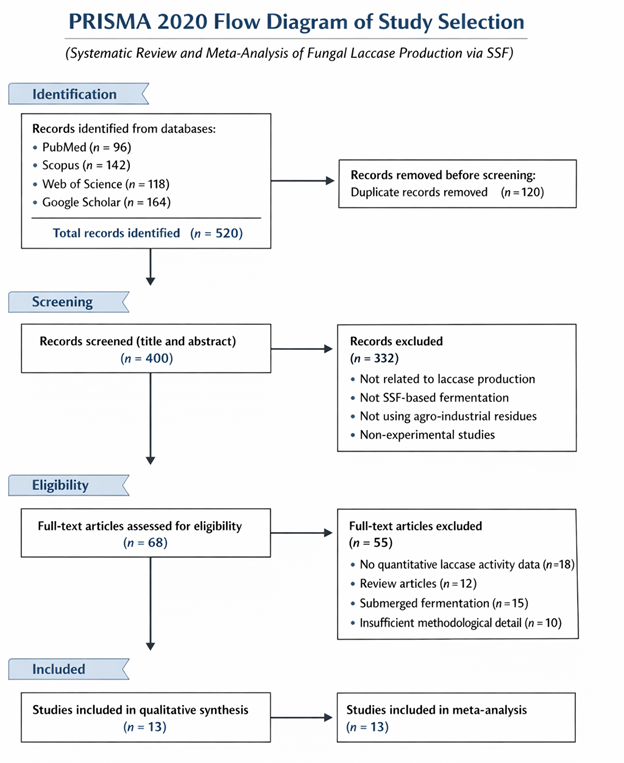
Given the rapidly expanding body of research in this field, systematic synthesis of available evidence has become increasingly important. Systematic reviews and meta-analyses provide structured frameworks for evaluating diverse studies and identifying consistent trends across experimental systems. The PRISMA guidelines, widely adopted in scientific literature, offer standardized procedures for reporting such reviews and ensuring transparency in study selection and data analysis (Urrútia & Bonfill, 2010). These methodological approaches are particularly valuable for assessing complex biotechnological systems, where multiple variables—such as substrate composition, fungal strain selection, and environmental conditions—can significantly influence outcomes.
The economic relevance of biomass-derived materials and enzymes further reinforces the need for integrated research efforts. Market analyses suggest that demand for cellulose-based fibers and related biomaterials is expected to grow substantially as industries transition toward renewable and biodegradable materials (Grand View Research, 2016). Similarly, enzyme markets continue to expand as biotechnology becomes increasingly central to sustainable industrial production.
Despite these advances, several challenges remain in optimizing laccase production through SSF. Variability in substrate composition, differences among fungal strains, and limitations in large-scale fermentation systems can all affect enzyme yields and process stability. Consequently, a comprehensive understanding of these variables is essential for developing efficient and scalable biotechnological processes.
In this context, the present systematic review aims to synthesize current knowledge regarding sustainable fungal laccase production through solid-state fermentation using agro-industrial residues. By examining substrate selection, fermentation conditions, fungal strains, and process optimization strategies, this review seeks to clarify key factors influencing enzyme production and highlight opportunities for integrating SSF technologies into circular bioeconomy frameworks. Through this analysis, the study contributes to the broader goal of developing sustainable biotechnological approaches that simultaneously address environmental challenges and industrial resource demands.