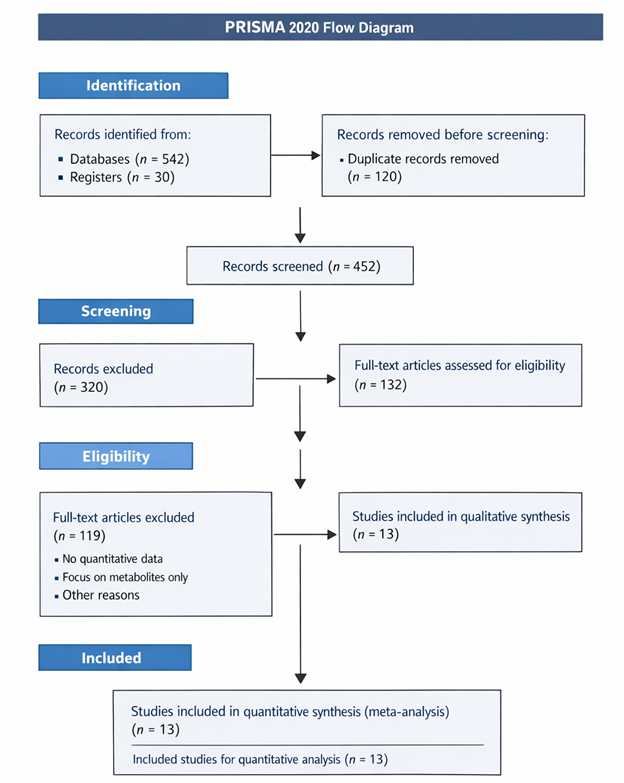
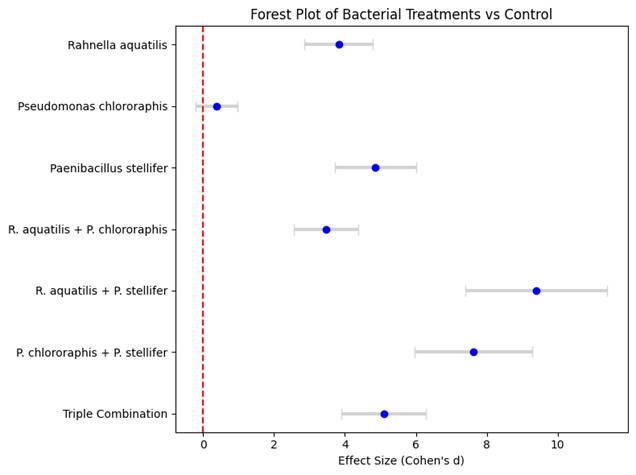
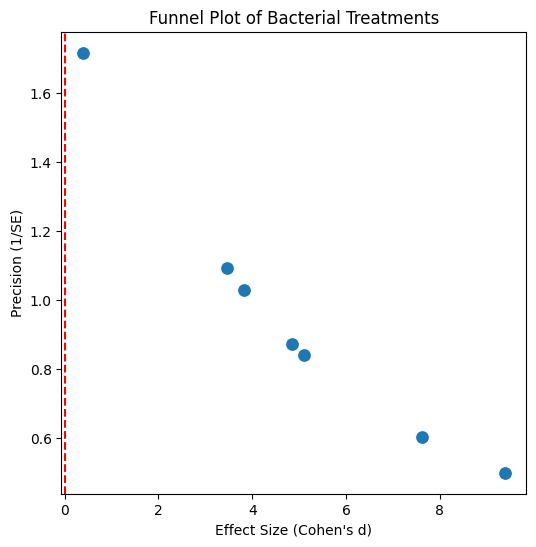
1.Introduction
Marine ecosystems harbor an immense and still only partially understood diversity of microorganisms that have shaped Earth’s biogeochemical cycles for billions of years. Among these microbial pioneers, cyanobacteria occupy a particularly intriguing position. These photosynthetic prokaryotes, once broadly referred to as blue-green algae, are believed to have emerged early in the evolutionary history of life and played a decisive role in transforming the planet’s atmosphere through oxygenic photosynthesis. Geological evidence suggests that the metabolic activity of ancient cyanobacteria contributed to the rise of atmospheric oxygen during the Precambrian era, thereby enabling the evolution of aerobic organisms and complex ecosystems (Bekker et al., 2004). Although this transformation occurred billions of years ago, the ecological and technological relevance of cyanobacteria continues to expand in the present day.
Cyanobacteria are remarkably adaptable organisms capable of colonizing a wide spectrum of habitats, from open oceans and freshwater lakes to terrestrial soils and extreme environments. Their resilience, combined with metabolic flexibility, allows them to persist in environments characterized by fluctuations in temperature, salinity, and nutrient availability. Indeed, cyanobacterial communities have been reported in environments ranging from polar ecosystems to hypersaline and thermal habitats, underscoring their extraordinary ecological versatility (Comte et al., 2007). In marine systems, these microorganisms frequently form intricate associations with other organisms, including sponges, algae, and invertebrates. Such symbiotic interactions can create highly specialized microenvironments that facilitate nutrient exchange, enhance microbial stability, and support complex ecological networks within marine ecosystems (Freeman & Thacker, 2011).
Beyond their ecological significance, cyanobacteria have increasingly drawn attention as valuable resources for biotechnology and sustainable development. Their photosynthetic capabilities allow them to convert solar energy and carbon dioxide into biomass and a diverse array of metabolites, offering a renewable platform for the production of bioactive compounds and industrial materials. Early investigations into cyanobacterial biotechnology highlighted their potential for generating biofuels, bioplastics, and specialty chemicals, suggesting that these organisms could serve as sustainable alternatives to fossil-based production systems (Abed et al., 2009). Advances in metabolic engineering have further expanded these possibilities by enabling the design of cyanobacterial strains capable of synthesizing high-value products, ranging from pharmaceuticals to bioenergy precursors (Ducat et al., 2011).
Among the most compelling applications of cyanobacteria is their role in renewable energy production. Certain strains have demonstrated the capacity to generate hydrogen through photobiological processes under aerobic conditions, illustrating their potential as environmentally friendly energy platforms (Bandyopadhyay et al., 2010). Similarly, industrial initiatives have explored the feasibility of using engineered cyanobacteria to produce bioethanol and other biofuels directly from sunlight and carbon dioxide. Such approaches align with broader efforts to develop carbon-neutral energy systems and reduce reliance on conventional fossil fuels (Algenol, 2016). Although these technologies remain under active development, they highlight the versatility of cyanobacteria as biological factories capable of contributing to a more sustainable energy landscape.
Another dimension of cyanobacterial importance lies in their remarkable capacity to synthesize structurally diverse secondary metabolites. Marine cyanobacteria, in particular, have been recognized as prolific sources of natural products with potent biological activities. Many of these compounds arise from specialized biosynthetic pathways, including non-ribosomal peptide synthases and polyketide synthases, which generate molecules with unique chemical architectures and pharmacological properties (Burja et al., 2001). Research into marine cyanobacterial metabolites has led to the discovery of numerous compounds with promising therapeutic potential. For instance, apratoxins isolated from cyanobacterial strains have shown strong anticancer activity, demonstrating the capacity of these metabolites to inhibit tumor cell proliferation (Luesch et al., 2002). Likewise, gallinamide A represents another example of a bioactive peptide with antimalarial potential, further illustrating the pharmaceutical promise of marine cyanobacteria (Linington et al., 2009).
The biochemical diversity of cyanobacteria is not limited to therapeutic compounds. These microorganisms also produce a wide array of pigments and protective molecules that enable them to thrive in challenging environmental conditions. Pigments such as chlorophylls, carotenoids, and other photoprotective compounds allow cyanobacteria to efficiently capture light energy while minimizing damage from ultraviolet radiation. In addition, microbial pigments like melanin have attracted growing interest due to their protective properties and potential applications in medicine, cosmetics, and environmental technologies (Guo et al., 2023). These molecules not only enhance the survival of cyanobacteria in exposed habitats but also represent valuable resources for industrial innovation.
However, the ecological success of cyanobacteria is accompanied by certain environmental challenges. Under conditions of nutrient enrichment, particularly elevated concentrations of phosphorus and nitrogen, cyanobacterial populations can proliferate rapidly and form harmful algal blooms. These events, often referred to as CyanoHABs, can produce toxins that threaten aquatic ecosystems, drinking water quality, and public health (Carmichael, 2001). Decades of research have documented the increasing occurrence of toxic cyanobacterial blooms worldwide, highlighting the need for improved monitoring and management strategies (Carmichael, 2008). Despite these concerns, understanding the mechanisms underlying cyanobacterial toxin production has also provided valuable insights into microbial ecology and metabolic regulation.
In marine environments, cyanobacteria frequently interact with macroalgae and other microorganisms, forming complex ecological partnerships. Red seaweeds belonging to the Rhodophyta, for example, are known to coexist with diverse microbial communities and produce nutraceutical compounds with antioxidant and therapeutic properties (Cotas et al., 2020). Such interactions often facilitate nutrient exchange and may enhance the stability of coastal ecosystems exposed to environmental stress. The coexistence of cyanobacteria with other marine organisms therefore represents a dynamic interface where ecological functions and biochemical innovation intersect.
In recent years, the integration of advanced analytical technologies has significantly accelerated research into cyanobacterial biology and metabolite discovery. Techniques such as Raman spectroscopy provide powerful, non-invasive tools for analyzing cellular components, enabling researchers to monitor intracellular structures and metabolic processes in living cells (Allakhverdiev et al., 2022). Complementary approaches, including metabolomics and functional genomics, have further expanded our understanding of cyanobacterial metabolic pathways and biosynthetic capabilities. For instance, untargeted metabolomic studies have revealed previously unknown secondary metabolites, demonstrating the immense chemical diversity present within cyanobacterial genomes (Baran et al., 2013). These technologies are rapidly transforming the exploration of marine microorganisms and facilitating the identification of novel bioactive compounds.
Analytical imaging methods have also enabled the visualization of intracellular structures such as lipid droplets and pigment distributions in microalgal cells. Label-free spectroscopic techniques, for example, allow researchers to investigate metabolic dynamics in living cells without the need for chemical staining, thereby preserving cellular integrity during analysis (Jaeger et al., 2016). Such tools provide valuable insights into cellular physiology and contribute to the optimization of microbial systems for biotechnology and biofuel production.
Beyond their roles in medicine and industry, cyanobacteria have begun to influence innovative approaches to sustainable infrastructure and environmental design. One notable example is the Bio-Intelligent Quotient (BIQ) building in Hamburg, which incorporates microalgae into architectural facades to capture carbon dioxide and generate renewable energy. This project demonstrates how microbial biotechnology can be integrated into urban environments to support climate-responsive architecture and resource efficiency (IBA Hamburg, 2013). These developments illustrate the expanding intersection between microbial ecology, engineering, and sustainable design.
Collectively, these diverse lines of research highlight the growing recognition of cyanobacteria as multifunctional organisms with profound ecological and technological significance. Their evolutionary history, metabolic versatility, and capacity to produce biologically active molecules position them as promising agents for addressing challenges related to food security, environmental sustainability, and human health. In agricultural contexts, cyanobacteria and associated beneficial microbes have gained attention as potential biofertilizers capable of enhancing plant growth, improving soil fertility, and reducing dependence on synthetic chemical inputs. For instance, the production of auxin-like compounds such as indole-3-acetic acid can facilitate root colonization and promote plant development, demonstrating the cross-kingdom influence of microbial metabolites (Hussain et al., 2015).
Given the increasing demand for sustainable agricultural practices and environmentally friendly bio-products, exploring the potential of marine cyanobacteria and their associated microbial communities has become an important research priority. This review therefore aims to synthesize current knowledge regarding the ecological roles, metabolic capabilities, and practical applications of marine cyanobacteria and beneficial microbes. By integrating insights from microbiology, biotechnology, and environmental science, the present work seeks to highlight emerging opportunities for harnessing these microorganisms in sustainable agriculture and a wide range of bio-applications.