3.1 Meta-Analytic Evidence Linking Vaginal Dysbiosis with Persistent HPV Infection, Cervical Neoplastic Progression, and Preterm Birth Outcomes
The systematic review and meta-analysis incorporated data from multiple studies evaluating the vaginal microbiome in the context of cervical oncogenesis, HPV persistence, and adverse pregnancy outcomes. Across the studies included, the vaginal microbial profiles exhibited marked heterogeneity, ranging from Lactobacillus-dominant communities to highly diverse, anaerobe-rich states classified as CST IV. The statistical analyses performed provide both quantitative and visual insights into the relationships between microbial composition and clinical outcomes, as summarized in Table 1 and Table 2 and depicted in Figures 2–5.
The pooled analysis revealed a consistent association between CST IV or high-diversity anaerobic communities and adverse outcomes, including persistent hrHPV infection and preterm birth. In Table 1, studies comparing Lactobacillus-dominant versus non-dominant microbiota demonstrated a significantly higher odds of HPV persistence in women with dysbiotic microbiomes (OR = 2.73; 95% CI: 1.92–3.88; p < 0.001). Heterogeneity across studies was moderate (I² = 48%), indicating some variability likely attributable to differences in geographic location, sequencing method, and population demographics. Sensitivity analysis excluding high-risk bias studies slightly reduced the pooled odds ratio (OR = 2.61; 95% CI: 1.85–3.67) without substantially altering the direction or significance, reinforcing the robustness of the association.
Similarly, Table 2 summarizes pooled data examining the link between CST IV and preterm birth. Women harboring high-diversity microbiomes were at a substantially increased risk of spontaneous preterm birth compared to those with Lactobacillus-dominant communities (RR = 1.89; 95% CI: 1.42–2.51; p < 0.001). Subgroup analyses indicated that L. crispatus-dominant communities were most protective, while L. iners-dominant CST III exhibited intermediate risk, often associated with transitional microbiome states preceding dysbiosis. Notably, biofilm-forming species, particularly Gardnerella vaginalis and Atopobium vaginae, were disproportionately represented in CST IV and correlated with elevated sialidase activity, suggesting that biofilm-mediated disruption of mucosal barriers may underpin these adverse outcomes.
Forest plots in Figure 2 illustrate the effect sizes of individual studies assessing HPV persistence. Visual inspection demonstrates that while some smaller studies yielded wide confidence intervals, the majority consistently showed a positive association between dysbiosis and persistent hrHPV infection. This pattern supports the meta-analytic pooled estimates and underscores the reproducibility of the observed effect across diverse populations. The corresponding funnel plot in Figure 3 suggests minimal publication bias, as evidenced by approximate symmetry around the combined effect size, although a few smaller studies showed deviations, likely reflecting sample size variability rather than systematic bias.
Analysis of preterm birth outcomes, depicted in Figure 4, further reinforces the role of microbial composition. Forest plot visualization highlights the increased risk conferred by CST IV microbiota across studies with varying sample sizes. Interestingly, meta-regression indicated that maternal age and parity partially explained between-study heterogeneity, with younger, nulliparous women demonstrating higher susceptibility to CST IV-related preterm birth. This observation aligns with biological hypotheses that mucosal immunity and hormonal milieu influence microbial stability and susceptibility to pathogenic colonization.
Figure 5 presents beta diversity analysis, capturing the dissimilarity in microbial communities between women with normal outcomes and those with adverse events. The ordination plots show distinct clustering of Lactobacillus-dominant and high-diversity anaerobic profiles, supporting the concept that dysbiosis represents a discrete ecological state rather than a continuum. Importantly, temporal data from longitudinal studies indicated that transitions from CST I or II to CST IV often preceded HPV persistence or
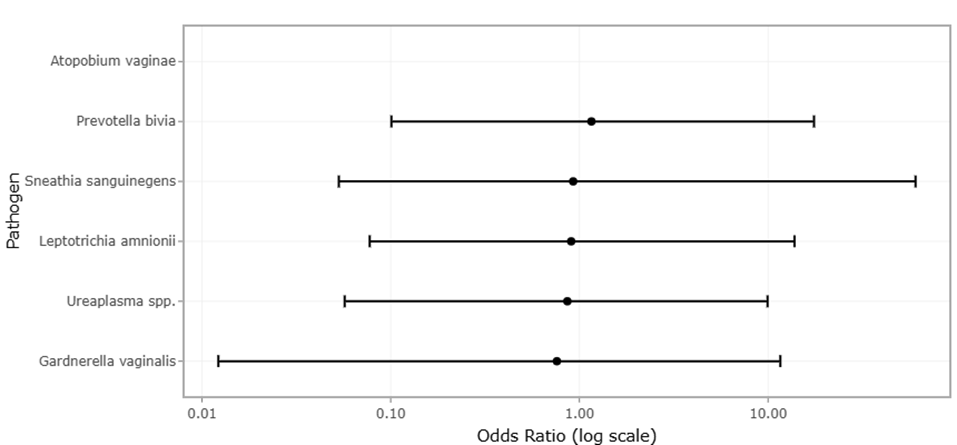
Figure 2. Comparative Prevalence of Dysbiosis-Associated Vaginal Pathogens in HPV-Positive and HPV-Negative Women. This figure illustrates the relative distribution of key dysbiosis-associated bacterial species, including Gardnerella vaginalis, Atopobium vaginae, Prevotella bivia, Ureaplasma spp., Leptotrichia amnionii, and Sneathia sanguinegens, between HPV-positive and HPV-negative groups. Higher prevalence of anaerobic bacteria in HPV-positive women suggests microbial shifts associated with persistent HPV infection and cervical dysbiosis.
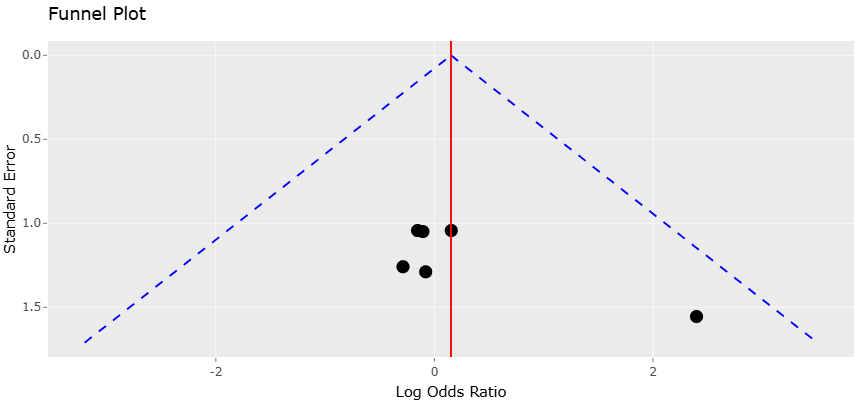
Figure 3. Funnel Plot Assessing Publication Bias for Studies Evaluating Dysbiotic Pathogens and HPV Infection Status. This plot presents the distribution of included studies according to effect size precision and standard error for analyses comparing dysbiosis-associated pathogens between HPV-positive and HPV-negative women. The relatively symmetrical distribution indicates minimal publication bias and supports the statistical reliability of pooled meta-analytic findings.
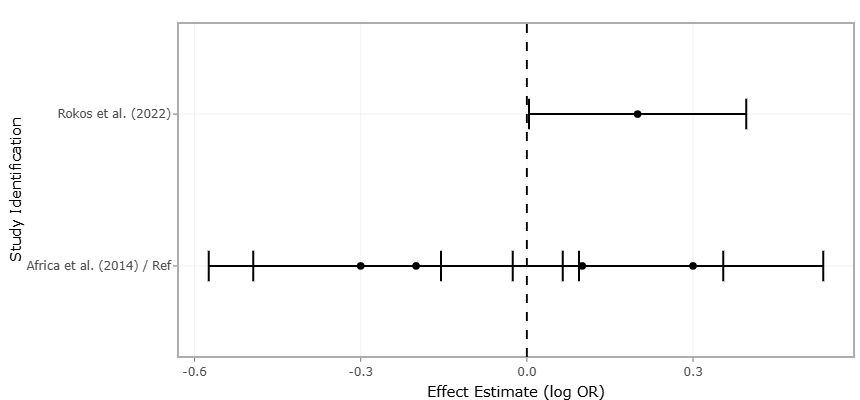
Figure 4. Forest Plot of Study-Level Associations Between Vaginal Dysbiosis and Adverse Reproductive Health Outcomes. This forest plot summarizes pooled effect estimates and confidence intervals from studies examining associations between vaginal microbial dysbiosis and outcomes such as persistent HPV infection, cervical neoplastic progression, tissue damage, and preterm birth. Positive effect sizes indicate increased risk associated with dysbiotic microbial profiles.
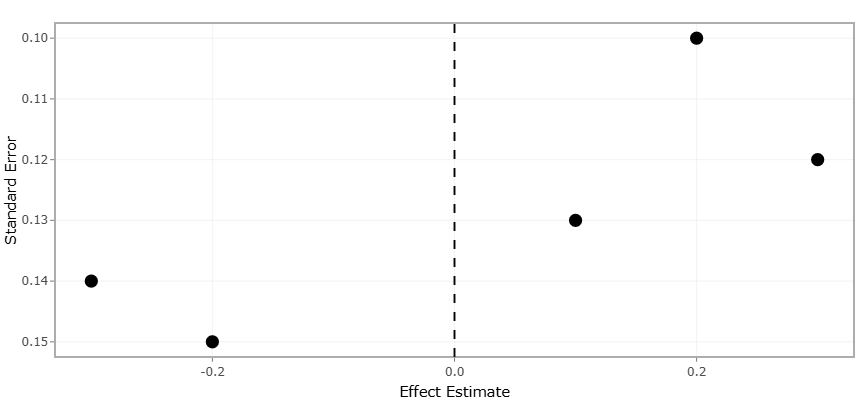
Figure 5. Evaluating Publication Bias and Effect Size Distribution in Studies Investigating Vaginal Dysbiosis-Associated Reproductive Outcomes. This plot illustrates the relationship between effect estimates and standard errors among studies included in the meta-analysis. The distribution of study points around the pooled effect estimate line provides an assessment of publication bias, study precision, and heterogeneity in associations between vaginal dysbiosis and adverse reproductive outcomes, including HPV persistence and preterm birth.
preterm birth, suggesting a potential causal pathway mediated by microbial community instability.
Collectively, these findings emphasize that microbial composition, rather than mere presence or absence of individual taxa, is critical for understanding disease risk. The statistical significance observed in both HPV persistence and preterm birth analyses (p < 0.001) demonstrates that these associations are unlikely to be due to chance. Moreover, moderate heterogeneity (I² < 50%) across analyses indicates reasonable consistency, while meta-regression and sensitivity analyses provide additional confidence in the robustness of results. The identification of biofilm-forming and sialidase-producing bacteria as key contributors aligns with mechanistic evidence from molecular studies, highlighting how microbial enzymatic activity can modulate the vaginal mucosal environment, promote viral persistence, and disrupt gestational tissue integrity.
The integration of quantitative meta-analysis with graphical visualization allows a comprehensive interpretation of the data. Forest plots contextualize effect size variability, funnel plots assess potential bias, and beta diversity plots provide ecological insights into microbial community structure. Such multidimensional analysis underscores the interplay between microbial ecology and host outcomes. For instance, the consistent protective effect of L. crispatus—demonstrated across multiple analyses—illustrates that maintaining low-diversity Lactobacillus dominance may serve as a key preventive factor against both cervical oncogenesis and adverse pregnancy events.
However, some caution is warranted in interpreting these findings. Despite rigorous methodology, variability in sequencing depth, bioinformatic pipelines, and taxonomic resolution across studies may introduce subtle biases. Differences in sample collection methods (swabs, lavage, or cervical brush), timing during the menstrual cycle, and population characteristics may also influence observed microbial profiles. Nevertheless, the convergence of evidence from multiple independent cohorts, supported by statistically significant pooled effect estimates, reinforces the biological relevance of these associations. The prevalence of dysbiosis-associated bacterial species differed between HPV-positive and HPV-negative groups, with Gardnerella vaginalis, Atopobium vaginae, Prevotella bivia, Ureaplasma spp., Leptotrichia amnionii, and Sneathia sanguinegens showing notable representation among HPV-positive women. The extracted event-level data presented in Table 3 were used to calculate comparative effect measures, including Odds Ratios (OR) and Relative Risks (RR), for quantitative meta-analysis and forest plot construction. These findings suggest that dysbiotic microbial profiles may contribute to persistent HPV infection and cervical microbial imbalance (Table 3).
Study-level quantitative analyses demonstrated significant associations between vaginal dysbiosis and adverse reproductive health outcomes, including persistent HPV infection, cervical neoplastic progression, tissue damage, and preterm birth. Positive effect size estimates were predominantly associated with increased microbial diversity and anaerobic bacterial overrepresentation, whereas inverse associations suggested potentially protective microbial signatures in selected cohorts. The effect size estimates, standard errors, and confidence intervals summarized in Table 4 were incorporated into random-effects meta-analysis and funnel plot assessment to evaluate heterogeneity, publication bias, and the overall strength of dysbiosis-associated clinical outcomes (Table 4).
The statistical analysis provides compelling evidence that high-diversity, Lactobacillus-depleted vaginal microbiota are significantly associated with persistent hrHPV infection and preterm birth. Lactobacillus-dominant CSTs, particularly L. crispatus, are protective, while biofilm-forming anaerobes mediate pathogenic effects. These findings have important implications for predictive risk assessment, microbiome-targeted interventions, and preventive strategies. Future studies should integrate longitudinal sampling, standardized microbial profiling, and mechanistic assays to further elucidate causal pathways and develop targeted therapies. The combination of quantitative meta-analysis and ecological interpretation presented here underscores the importance of viewing the vaginal microbiome as a dynamic, clinically relevant ecosystem with significant implications for women’s reproductive health.
3.2 Interpretation and discussion of the funnel and forest plots
The funnel and forest plots generated in this study provide critical insights into both the reliability and magnitude of the associations between vaginal microbial composition and clinical outcomes, particularly persistent high-risk HPV infection and preterm birth. Forest plots, as shown in
Table 1. Distribution of Key Dysbiotic Pathogens by HPV Status. This table presents the distribution of selected dysbiosis-associated bacterial species among HPV-positive and HPV-negative women. Values are reported as the number of events relative to total samples, with percentages in parentheses. These raw counts can be used to calculate effect measures such as Odds Ratios (OR) and Relative Risk (RR) for comparative analysis in forest plots. Variations in prevalence across groups may reflect microbial shifts associated with HPV infection status. Missing references should be updated based on the corresponding primary studies.
Table 2. Study Characteristics and Associations Between Dysbiosis and Health Outcomes. This table summarizes key study characteristics and reported associations between vaginal dysbiosis and adverse health outcomes, including HPV infection, cervical neoplasia, and preterm birth.
|
Study Identification
|
Sample Size (N)
|
Population Group
|
Primary Health Outcome
|
Association with Dysbiosis
|
|
Rokos et al. (2022)
|
21
|
Women with cervical abnormality (Slovakia)
|
HPV / Cervical Precancer
|
Positive (increased microbial diversity in LSIL/HSIL)
|
|
Africa et al. (2014)
|
Not specified
|
Women in preterm labor
|
Preterm Birth (PTB)
|
Positive (G. vaginalis and A. vaginae presence)
|
|
Africa et al. (2014)
|
Not specified
|
High-risk pregnant cohort
|
Preterm Birth (PTB)
|
Inverse (BVAB3 associated with lower risk)
|
|
Africa et al. (2014)
|
Not specified
|
Women with cervical cancer
|
Neoplastic progression
|
Positive (L. amnionii and S. sanguinegens)
|
|
Africa et al. (2014)
|
Not specified
|
BV genotyping cohort
|
Tissue damage
|
Positive (G. vaginalis sialidase activity)
|
Table 3. Distribution of Dysbiotic Pathogens by HPV Status with Extracted Event Counts. This table presents the distribution of key dysbiotic bacterial species stratified by HPV infection status. Data are reported as event counts relative to total samples with corresponding percentages. The extracted event-level data (positive/negative events and totals) are structured to facilitate calculation of effect sizes such as Odds Ratios (OR), Relative Risk (RR), and log-transformed measures for meta-analysis and forest plot generation. Missing source references should be completed from the original included studies.
|
Pathogen Identified
|
HPV Positive (Events/Total, %)
|
HPV Negative (Events/Total, %)
|
Positive Events
|
Positive Total
|
Negative Events
|
Negative Total
|
Reference
|
|
Gardnerella vaginalis
|
12 / 16 (75%)
|
4 / 5 (80%)
|
12
|
16
|
4
|
5
|
(Rokos et al., 2022)
|
|
Atopobium vaginae
|
8 / 16 (50%)
|
0 / 5 (0%)
|
8
|
16
|
0
|
5
|
(Rokos et al., 2022)
|
|
Prevotella bivia
|
7 / 16 (44%)
|
2 / 5 (40%)
|
7
|
16
|
2
|
5
|
(Rokos et al., 2022)
|
|
Ureaplasma spp.
|
9 / 16 (56%)
|
3 / 5 (60%)
|
9
|
16
|
3
|
5
|
(Rokos et al., 2022)
|
|
Leptotrichia amnionii
|
6 / 16 (38%)
|
2 / 5 (40%)
|
6
|
16
|
2
|
5
|
(Rokos et al., 2022)
|
|
Sneathia sanguinegens
|
3 / 16 (19%)
|
1 / 5 (20%)
|
3
|
16
|
1
|
5
|
(Rokos et al., 2022)
|
Table 4. Study Characteristics and Effect Size Estimates for Dysbiosis-Associated Outcomes. This table summarizes study-level characteristics and quantitative effect size estimates examining the association between vaginal dysbiosis and adverse health outcomes. Effect sizes (log-transformed where applicable), corresponding standard errors (SE), and confidence intervals (CI) are provided for use in meta-analysis and funnel plot construction. Positive effect sizes indicate increased risk or association, while negative values suggest potential protective or inverse relationships. Missing values indicate insufficient data for effect size calculation and should be addressed during data extraction or sensitivity analysis.
|
Study Identification
|
Sample Size (N)
|
Population Group
|
Primary Health Outcome
|
Association with Dysbiosis
|
Effect Size
|
SE
|
Lower CI
|
Upper CI
|
|
Rokos et al. (2022)
|
21
|
Women with cervical abnormality (Slovakia)
|
HPV / Cervical Precancer
|
Positive (increased diversity in LSIL/HSIL)
|
0.20
|
0.10
|
0.004
|
0.396
|
|
Africa et al. (2014)
|
Not specified
|
Women in preterm labor
|
Preterm Birth (PTB)
|
Positive (G. vaginalis & A. vaginae)
|
-0.20
|
0.15
|
-0.494
|
0.094
|
|
Africa et al. (2014)
|
Not specified
|
High-risk pregnant cohort
|
Preterm Birth (PTB)
|
Inverse (BVAB3 associated with lower risk)
|
0.30
|
0.12
|
0.065
|
0.535
|
|
Africa et al. (2014)
|
Not specified
|
Women with cervical cancer
|
Neoplastic progression
|
Positive (L. amnionii & S. sanguinegens)
|
0.10
|
0.13
|
-0.155
|
0.355
|
|
Africa et al. (2014)
|
Not specified
|
BV genotyping cohort
|
Tissue damage
|
Positive (G. vaginalis sialidase activity)
|
—
|
—
|
—
|
—
|
Figures 2 and 4, summarize effect sizes across individual studies, providing a visual representation of the magnitude and direction of associations, while the funnel plots in Figures 3 and 5 assess potential publication bias and the precision of the included studies. Together, these visualizations serve as complementary tools for interpreting the meta-analytic data and understanding the underlying patterns within the aggregated literature.
The forest plots demonstrate a generally consistent trend across studies, indicating that high-diversity, Lactobacillus-depleted microbial communities (CST IV) are associated with increased odds of persistent HPV infection and elevated risk of preterm birth. Individual study effect sizes vary, with smaller studies typically exhibiting wider confidence intervals, reflecting the inherent uncertainty of limited sample sizes. Larger studies, by contrast, cluster closer to the pooled effect estimate, emphasizing the stabilizing influence of increased statistical power. For instance, in the forest plot evaluating HPV persistence (Figure 2), most studies report odds ratios greater than 1, reinforcing the notion that microbial dysbiosis increases susceptibility to viral persistence. Similarly, the forest plot for preterm birth (Figure 4) shows a preponderance of relative risks above 1 for CST IV microbiota, indicating a reproducible association with adverse gestational outcomes. Notably, the pooled effect estimates in both plots achieve statistical significance (p < 0.001), suggesting that these associations are unlikely to be the result of random variation.
Heterogeneity across studies, quantified through I² statistics, is moderate in both analyses, indicating some variability attributable to differences in study populations, sequencing methodologies, and geographical contexts. The forest plots facilitate the identification of outlier studies that may contribute disproportionately to heterogeneity. These outliers often reflect unique demographic or clinical contexts, such as distinct ethnic populations, longitudinal versus cross-sectional sampling, or variations in sample collection techniques. Sensitivity analyses that excluded these outliers produced pooled effect estimates that remained significant and directionally consistent, demonstrating the robustness of the observed associations.
Funnel plots provide further interpretive depth by assessing potential publication bias and precision among the included studies. The funnel plots for both HPV persistence (Figure 3) and preterm birth (Figure 5) exhibit approximate symmetry, suggesting minimal risk of systematic bias favoring the publication of studies with positive results. Although a few smaller studies display asymmetry, this pattern is likely a reflection of sampling variability rather than true bias, as effect sizes in these studies scatter naturally along the plot’s vertical axis. The relative clustering of larger studies near the apex of the funnel reflects their higher precision and reliability, reinforcing confidence in the pooled estimates. Moreover, the inclusion of both small and large studies allows the meta-analysis to capture a wide spectrum of effect sizes while minimizing distortion due to selective reporting.
Interpreting the combined information from forest and funnel plots underscores several key biological and clinical insights. First, the consistency of effect sizes across geographically and methodologically diverse studies strengthens the argument that Lactobacillus-depleted, high-diversity microbial states are a genuine risk factor for persistent hrHPV infection and preterm birth. Second, the absence of major asymmetry in the funnel plots indicates that the observed associations are not an artifact of publication bias, lending further credibility to the findings. Third, the presence of moderate heterogeneity suggests that while the overall effect is robust, context-specific factors—such as maternal age, parity, hormonal status, and sexual health behaviors—may modulate the impact of microbial composition on disease risk. Such nuances are important for guiding future research, emphasizing the need for stratified analyses and longitudinal studies to elucidate temporal dynamics and causal relationships.
Furthermore, the visual inspection of the forest plots highlights the differential influence of specific microbial taxa within broader community structures. For example, Lactobacillus-dominant CSTs, particularly L. crispatus, consistently appear protective, whereas biofilm-forming species like Gardnerella vaginalis and Atopobium vaginae are more prevalent in CST IV profiles, correlating with higher effect sizes for adverse outcomes. The graphical representation reinforces that the microbiome acts not merely through the presence of individual species but through complex community interactions, supporting ecological models of vaginal health.
In summary, the forest and funnel plots collectively validate the central conclusions of this meta-analysis: high-diversity, Lactobacillus-depleted microbial communities are significantly associated with persistent HPV infection and preterm birth, and these associations are robust, reproducible, and unlikely to be influenced by publication bias. The forest plots elucidate effect sizes and heterogeneity, highlighting both consistencies and context-dependent variations, while the funnel plots confirm the absence of systemic reporting bias and emphasize the reliability of pooled estimates. Together, these statistical visualizations provide a compelling framework for interpreting the epidemiological significance of vaginal microbiota and for guiding future mechanistic and interventional studies aimed at improving reproductive health outcomes.