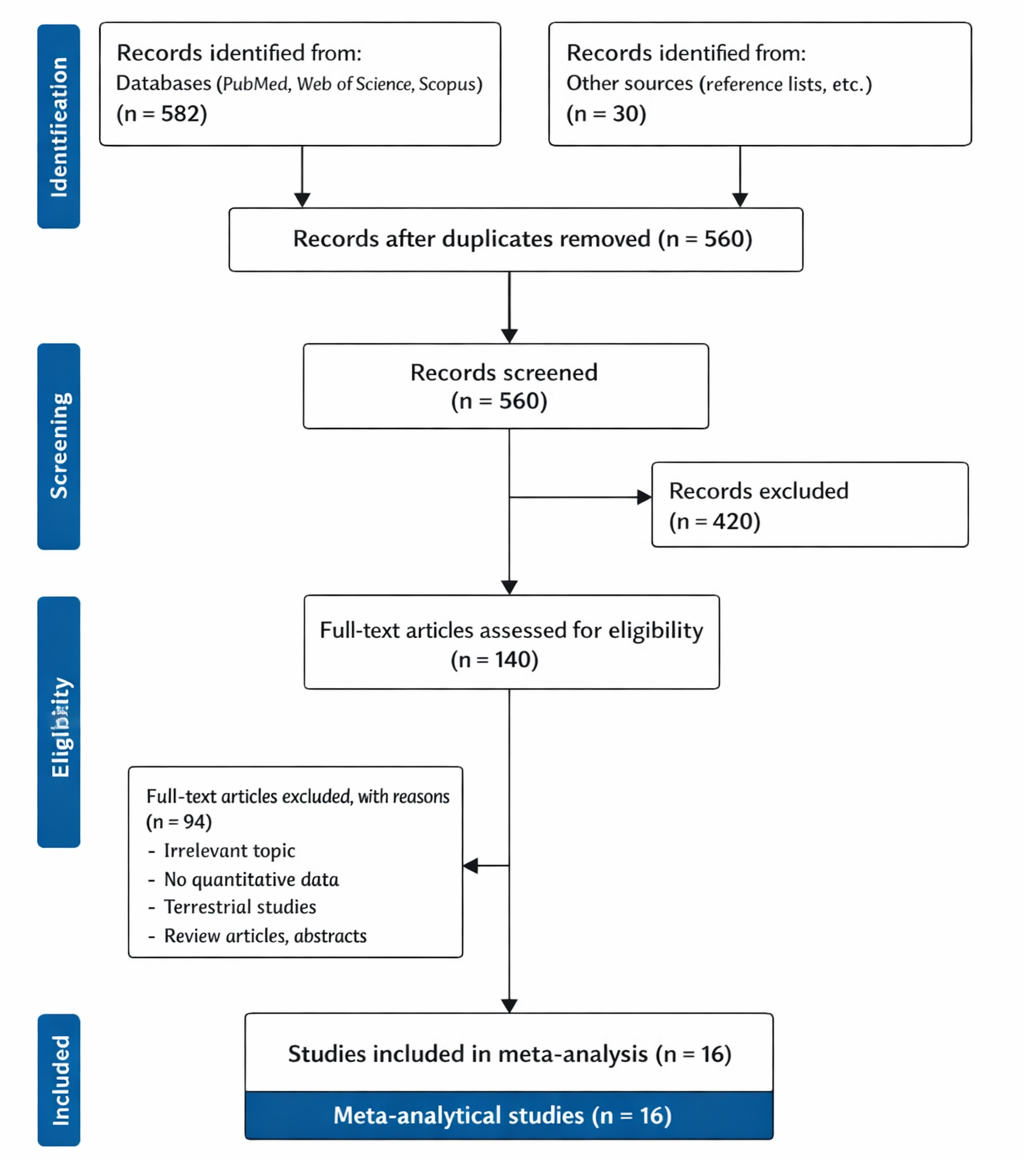
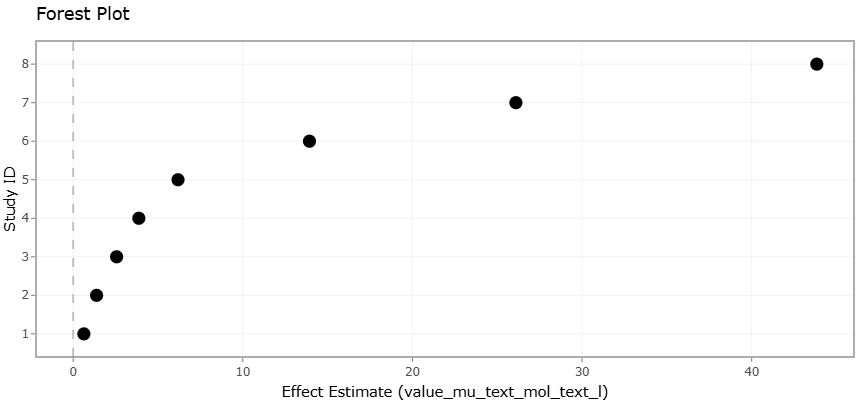
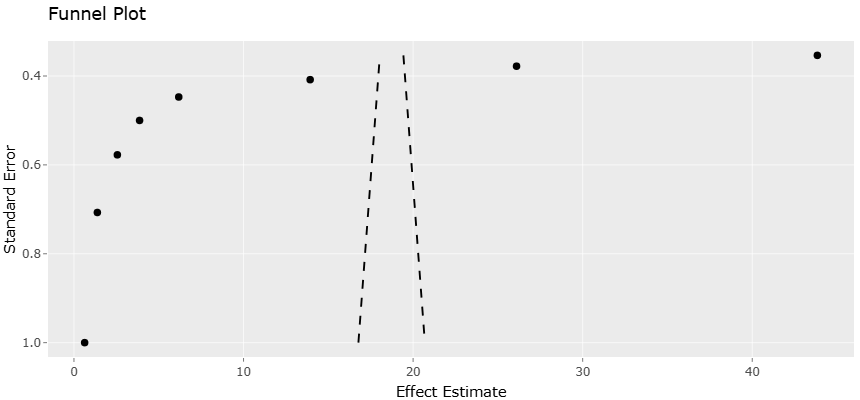
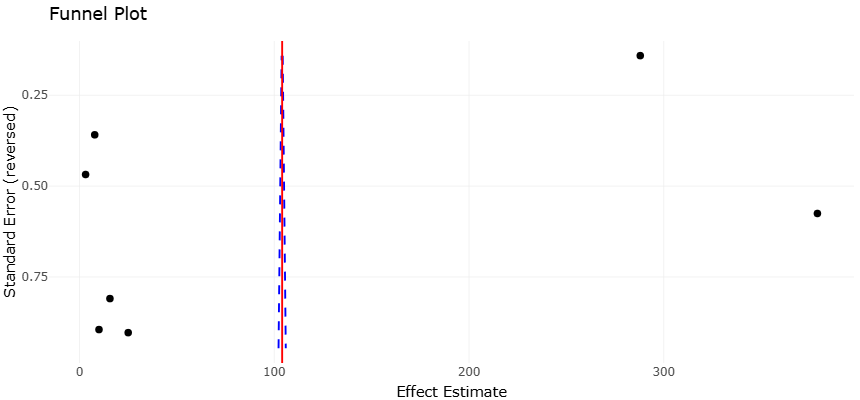
1. Introduction
Aquatic ecosystems are undergoing profound transformations driven by accelerating anthropogenic pressure and global climate change. Among the most visible and ecologically disruptive consequences of these changes is the intensification of water eutrophication, a process fueled by excessive nutrient loading from agriculture, urban runoff, and industrial activities. Eutrophication creates favorable conditions for the proliferation of harmful cyanobacteria, leading to recurrent and often persistent cyanobacterial blooms in freshwater and coastal environments (Luo et al., 2021). These blooms disrupt food webs, deplete oxygen, degrade water quality, and pose direct risks to human and animal health through the production of potent cyanotoxins such as microcystins (Zhang et al., 2023). At the same time, global health systems are grappling with a parallel crisis: the rapid emergence and spread of antimicrobial resistance, driven largely by the extensive and often indiscriminate use of synthetic antibiotics (Zuorro et al., 2024). Together, these intertwined ecological and medical challenges underscore the urgent need for novel, sustainable, and environmentally compatible bioactive agents.
Conventional chemical algicides and antibiotics, while effective in the short term, are increasingly recognized as unsustainable solutions. Many exhibit low selectivity, persist in the environment, disrupt non-target organisms, and contribute to resistance development (El Amrani Zerrifi et al., 2018; Zuorro et al., 2024). In this context, marine ecosystems have emerged as an exceptionally rich yet still underexplored reservoir of biologically active compounds. Marine macroalgae, microalgae, cyanobacteria, and their associated microorganisms have evolved under intense competition for space, nutrients, and light, driving the diversification of sophisticated chemical defense and communication strategies (Beev et al., 2025). These organisms synthesize a wide array of secondary metabolites, including terpenoids, phenolic compounds, alkaloids, sulfated polysaccharides, and fatty acid derivatives, many of which exhibit antimicrobial, antiviral, antifungal, anti-inflammatory, and algicidal activities (El Amrani Zerrifi et al., 2018; Zuorro et al., 2024).
Over the past decade, systematic reviews and quantitative syntheses have increasingly highlighted the consistency and potency of marine-derived bioactive compounds across diverse experimental systems. Meta-analytical evaluations of algicidal and antimicrobial efficacy demonstrate that certain marine natural products rival or exceed the activity of conventional chemical controls, often at substantially lower concentrations (Luo et al., 2021; Zhang et al., 2023). Importantly, these compounds frequently display species-specific or pathway-targeted effects, suggesting a reduced likelihood of broad ecological disruption. Such findings position marine bioactives not merely as chemical curiosities, but as realistic candidates for next-generation environmental and biomedical applications.
Central to understanding the origin and function of many marine bioactive compounds is the concept of chemical communication. In the ocean, microorganisms do not exist as isolated entities; instead, they form dynamic, chemically mediated networks. One of the most intensively studied communication systems is quorum sensing (QS), a regulatory mechanism through which bacteria coordinate gene expression in response to local population density using small diffusible signaling molecules known as autoinducers (Dow, 2021). QS enables collective behaviors such as biofilm formation, motility regulation, virulence expression, and secondary metabolite production. In marine environments, QS takes on added ecological complexity because it frequently operates across species boundaries and even across kingdoms.
A particularly important hotspot for QS-mediated interactions is the phycosphere—the microscale region surrounding algal cells where dissolved organic matter released by algae creates a nutrient-rich niche for heterotrophic bacteria (Qiao et al., 2022). Within this microenvironment, bacteria and algae engage in tightly coupled interactions that range from mutualism to antagonism. QS signals such as N-acyl homoserine lactones (AHLs), autoinducer-2 (AI-2), alkylquinolones (AQs), and their derivatives regulate bacterial behaviors that directly influence algal growth, survival, and bloom dynamics (Dow, 2021; Qiao et al., 2022). Evidence synthesized across multiple studies indicates that these signals can function not only as communication cues but also as direct bioactive agents with algicidal or growth-modulating properties.
AHLs are among the most widely distributed QS signals in marine systems and are primarily produced by Gram-negative bacteria, including members of the ecologically prominent Roseobacter clade (Wagner-Döbler et al., 2005; Ziesche et al., 2015). Their structural diversity, driven by variations in acyl chain length and functional substitutions, translates into highly specific biological effects. Systematic analyses reveal that long-chain AHLs prevalent in the phycosphere can either stimulate algal growth or induce growth inhibition, depending on both algal species and AHL structure (Stock et al., 2020; Qiao et al., 2022). Notably, some AHLs undergo spontaneous chemical rearrangement to form tetramic acids, compounds that exhibit potent algicidal activity through the disruption of photosynthetic electron transport (Stock et al., 2019; Dow, 2021). These findings illustrate how QS chemistry can blur the boundary between signaling and chemical warfare.
Beyond AHLs, AI-2 functions as a more universal signal, produced by both Gram-positive and Gram-negative bacteria and detected in diverse marine microbial assemblages (Schauder et al., 2001; Pereira et al., 2013). Meta-analytical integration of experimental studies suggests that AI-2 plays a significant role in nutrient cycling rather than direct algicidal activity. For example, AI-2–mediated regulation of alkaline phosphatase activity in Trichodesmium consortia highlights its involvement in phosphorus acquisition under nutrient-limited conditions (Van Mooy et al., 2012; Hmelo et al., 2011). Such regulatory functions may indirectly shape bloom formation and decline by altering nutrient availability at the microscale.
Alkylquinolones represent another class of QS-associated compounds with pronounced ecological impact. While some AQs function as bona fide signaling molecules, many act directly as algicides. Quantitative syntheses show that compounds such as 2-heptyl-4-quinolone (HHQ) exert strong inhibitory effects on marine phytoplankton, including Emiliania huxleyi, by targeting photosystem II and the cytochrome b6f complex, as well as arresting cell cycle progression (Dow et al., 2020; Pollara et al., 2021). These effects, observed consistently across independent studies, support the notion that QS-regulated metabolites can serve as precise tools for controlling algal populations.
Importantly, algae are not passive participants in these chemically mediated interactions. Many species produce compounds that interfere with bacterial QS, a phenomenon known as quorum quenching (QQ). Red algae, for instance, release halogenated furanones that competitively inhibit AHL receptors, thereby preventing bacterial colonization and biofilm formation (Manefield et al., 1999; Hentzer et al., 2002). Diatoms such as Nitzschia cf. pellucida enzymatically degrade AHLs, neutralizing their signaling capacity (Syrpas et al., 2014). From a systems perspective, these QQ mechanisms highlight an evolutionary arms race in which chemical signaling and signal disruption co-evolve to shape microbial community structure.
Taken together, evidence synthesized through systematic review and meta-analysis underscores the central role of marine chemical ecology in governing algae–bacteria interactions and regulating ecosystem function. The consistency of observed bioactivities across taxa and experimental designs strengthens confidence in the translational potential of these compounds. By integrating ecological insight with advances in analytical chemistry, genomics, and co-culture methodologies, contemporary research is increasingly able to activate silent biosynthetic pathways and uncover novel metabolites that would otherwise remain hidden (Li et al., 2023). Such interdisciplinary approaches offer a pathway toward the rational development of marine-derived algicides and antimicrobials that are both effective and environmentally responsible.
In an era defined by ecological instability and diminishing efficacy of conventional antimicrobials, marine bioactive compounds and QS-mediated interactions offer not only a deeper understanding of oceanic microbial life but also a promising foundation for sustainable solutions in environmental management and human health.