1. Introduction
The pressure on global food systems is no longer theoretical—it is immediate and deeply structural. Agricultural systems are being asked to produce more food from increasingly constrained land, water, and nutrient resources. At the same time, continuous cropping, soil fatigue, disease emergence, and climate-induced stressors such as drought and salinity are weakening productivity and resilience (Zeeshan Ul Haq et al., 2023; Malik et al., 2022). These intensifying pressures have renewed interest in biological solutions capable of restoring soil fertility while sustaining yield. Increasingly, attention has turned toward beneficial soil microbiomes and their role in improving crop performance and ecological stability (Vincze et al., 2024).
Among these biological allies, fungi occupy a uniquely versatile position. Fungal diversity is vast, and environmental DNA studies continue to reveal that described species represent only a fraction of what exists (Wu et al., 2019). This immense biodiversity spans agricultural soils, extreme environments such as Antarctica, and specialized ecological niches like caves—habitats that are now being recognized as reservoirs of biotechnologically valuable metabolites (Zucconi et al., 2020; Barbosa et al., 2025). Such diversity suggests that fungal systems offer far more than traditional fermentation applications; they represent a largely untapped resource for sustainable innovation.
In soil ecosystems, fungi contribute fundamentally to nutrient cycling and fertility restoration. Compost-associated microbial communities improve soil structure, organic matter turnover, and nutrient bioavailability (Aguilar-Paredes et al., 2023). Mycorrhizal fungi, in particular, form symbiotic associations with plant roots, enhancing phosphorus and nitrogen acquisition and improving stress tolerance (Bortolot et al., 2024). Similarly, species of Mortierella and other plant growth-promoting fungi enhance nutrient mobilization and root development in agricultural soils (Ozimek & Hanaka, 2021). These interactions become especially important under environmental stress, where microbial symbioses can help maintain crop productivity.
Emerging research also highlights the importance of seed-associated and endophytic microbiomes. Seed endophytes function as early colonizers that can influence plant growth trajectories and stress resilience (Fadiji et al., 2025). Endophytes more broadly occupy dynamic ecological niches within plants, sometimes acting as permanent symbionts and at other times as context-dependent mutualists (Kuzniar et al., 2025). Their agricultural value becomes particularly visible under abiotic stress conditions; for example, microbial inoculation has been shown to enhance survival and nutritional quality in crops grown under water deficit (Almeida et al., 2024).
Beyond growth promotion, fungi contribute to soil remediation and nutrient biofortification. Native fungal strains are increasingly investigated as nature-based solutions to mitigate toxic metal(loid) accumulation in staple crops such as rice (Canonica et al., 2025). Likewise, beneficial microorganisms can enhance selenium biofortification in crops, addressing micronutrient deficiencies linked to human health disorders (Ye et al., 2020). Selenium-related nanobiotechnological approaches, including biologically derived nanoparticles, further illustrate how microbial systems intersect with agronomy and nutritional enhancement (Ciobanu et al., 2024). Additionally, actinomycetes and related rhizosphere organisms enrich nitrogen availability and improve seed quality in legumes, reinforcing the broader importance of microbial partnerships in sustainable production (AbdElgawad et al., 2020).
Fungi also contribute to circular agricultural systems through organic waste valorization. Insect-derived frass, for example, supports soil fertility and microbial proliferation when integrated into crop systems (Verardi et al., 2025). Through fermentation and metabolic transformation, fungal systems convert low-value substrates into protein-rich biomass and bioactive compounds, aligning with sustainability and waste reduction goals.
The nutritional dimension of fungal biotechnology is equally compelling. Edible and medicinal mushrooms contain bioactive compounds that modulate immune function and contribute to metabolic health (Zhao et al., 2020). Fungal metabolites—including polysaccharides, antioxidants, and secondary metabolites—have been associated with improved physiological resilience and functional food development (Dufossé et al., 2021). More recent analyses emphasize fungi’s broader role in improving human and animal life through diverse nutritional and therapeutic applications (Dufossé, 2024).
Mycoproteins derived from filamentous fungi represent one of the most visible examples of fungal contribution to sustainable diets. These protein-rich foods offer favorable amino acid profiles and have been linked to improved glycemic control, satiety, and cardiometabolic markers across the lifespan (Derbyshire, 2022). Meanwhile, fungal lipid metabolism enables the production of arachidonic acid–enriched oils and other polyunsaturated fatty acids through fermentation technologies (Slaný et al., 2020). Such innovations reduce dependence on conventional animal-derived sources while offering scalable nutritional alternatives.
Fungal metabolites also serve as natural pigments and bioactive compounds in food systems. Pigments derived from fungal fermentation provide antioxidant and antimicrobial benefits in addition to coloration (Lin & Xu, 2020). The metabolic versatility of fungi supports the development of functional foods aimed at promoting healthy aging and sustainability (Takahashi et al., 2020). These developments illustrate how fungal metabolism bridges nutrition, health, and industrial application.
Industrial enzyme production further reinforces fungal importance. Endophytic fungi produce proteases and other enzymes with applications in food processing and therapeutic contexts (Bezerra et al., 2021). Cold-adapted and extremophilic fungi continue to expand enzymatic possibilities, particularly in energy-efficient biocatalysis (Zucconi et al., 2020). In parallel, exploration of cave-derived fungi underscores the promise of understudied ecological niches as reservoirs of novel enzymatic systems (Barbosa et al., 2025).
Taken together, these lines of evidence point toward a coherent theme: fungi are not peripheral to sustainable development—they are central to it. They enhance soil fertility, mitigate environmental stress, improve nutrient density, contribute to food innovation, and generate high-value biomolecules across sectors. Yet despite this growing body of work, fungal systems remain underintegrated into mainstream agricultural and nutritional policy frameworks.
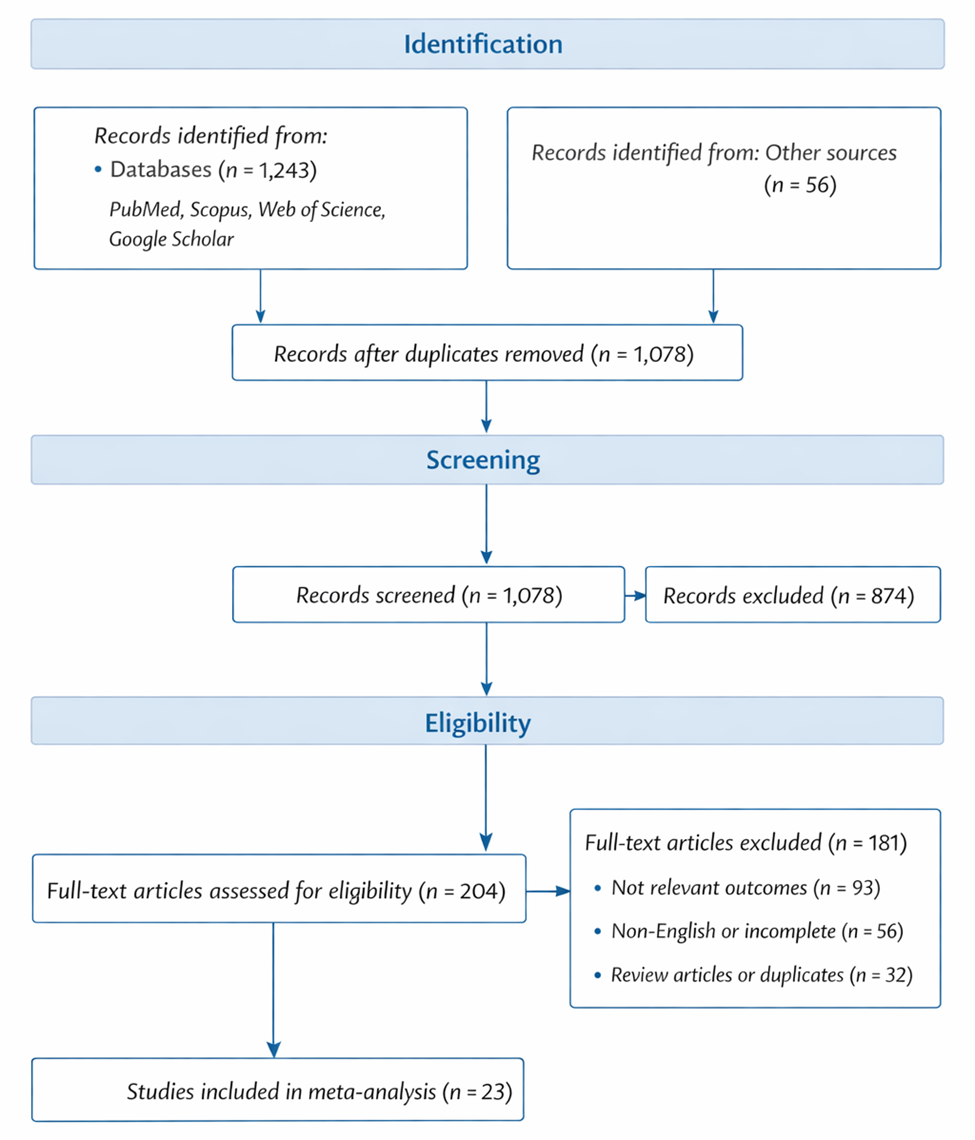
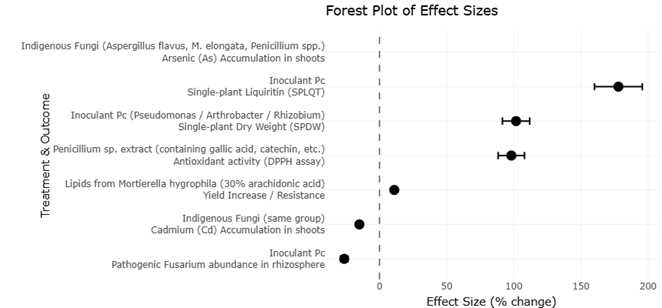
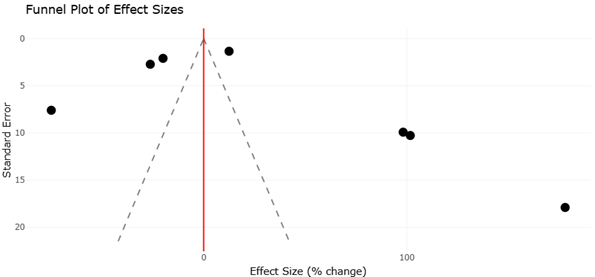
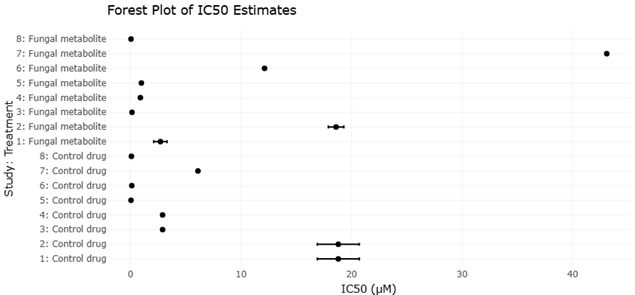
This systematic review and meta-analysis synthesizes current evidence across ecological, agronomic, nutritional, and biotechnological domains. By integrating findings from studies on soil microbiomes, mycorrhizal associations, endophytes, functional foods, enzyme production, biofortification, and waste valorization, we aim to clarify how fungi and their metabolites can support more resilient food systems. Rather than viewing fungi solely as decomposers or fermentation agents, this work frames them as multidimensional biological assets capable of contributing simultaneously to sustainability, nutrition, and human health.
In an era defined by climate instability, resource scarcity, and rising nutritional demands, solutions must be biologically intelligent and systemically integrated. Fungi—diverse, adaptable, metabolically versatile—offer precisely such a pathway forward.