1. Introduction
Fungi, perhaps more than most biological systems, occupy an uneasy yet fascinating position between nourishment and danger. On one hand, edible mushrooms have been woven into culinary traditions for centuries, valued not only for their distinct umami character but also for their emerging role as functional foods. On the other, certain species remain among the most toxic natural organisms known. This duality—almost paradoxical in nature—continues to shape scientific inquiry into fungal biology. Increasingly, research is moving beyond simple compositional descriptions, attempting instead to understand how biochemical complexity translates into nutritional benefit or, in some cases, life-threatening toxicity.
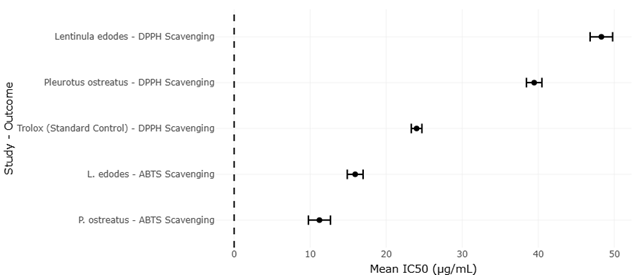
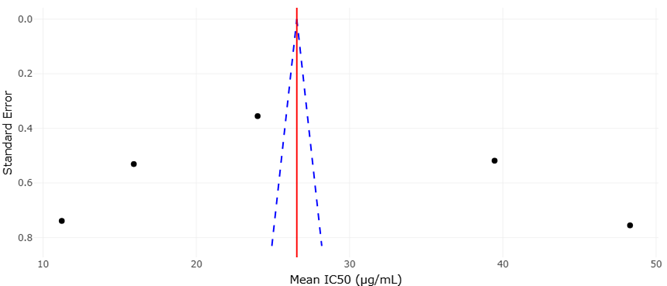
Edible mushrooms such as Hypsizygus marmoreus, Lentinula edodes, and Pleurotus ostreatus are frequently highlighted as exemplary models of this beneficial side of fungal metabolism. Their value extends well beyond macronutrient content, encompassing a wide array of bioactive molecules that contribute to antioxidant, immunomodulatory, and even anticancer effects (Rahi & Malik, 2016; Elhusseiny et al., 2021). Still, the appreciation of mushrooms as nutraceuticals is not entirely straightforward. Flavor, for instance, is not a fixed trait but rather an outcome of dynamic metabolic processes influenced by growth stage, environmental conditions, and genetic background (Cho et al., 2007; Politowicz et al., 2018).
In fact, even within a single fruiting body, compositional heterogeneity can be surprisingly pronounced. The cap and stipe—structures that may appear functionally similar at first glance—often diverge significantly in their biochemical profiles. Caps tend to accumulate higher levels of amino acids and secondary metabolites associated with taste and bioactivity, whereas stipes more frequently serve as repositories of structural carbohydrates (Park et al., 2017; Wang et al., 2018). Such spatial differentiation suggests that mushrooms are not uniform biological entities but rather metabolically compartmentalized systems, an idea that is gaining increasing attention in food science and metabolomics. A slightly more nuanced layer of variation also emerges when considering how maturity, strain specificity, and growth conditions collectively shape mushroom composition. Earlier work has shown that the stage of fruiting body development can significantly influence both antimicrobial activity and chemical profiles, with measurable shifts in bioactive compounds as maturation progresses (Barros et al., 2007). Similarly, differences in strain selection and cultivation substrates have been linked to variability in taste-related components, suggesting that metabolic output is not only developmentally regulated but also highly context-dependent (Harada et al., 2004). Complementing these findings, metabolite profiling approaches using mass spectrometry have further demonstrated that antioxidant capacity and biochemical composition can vary markedly across growth stages, reinforcing the idea that mushrooms represent dynamic biochemical systems rather than static nutritional entities (Lee et al., 2012).
Temporal variation adds another layer of complexity. During development, mushrooms undergo substantial metabolic reprogramming. Early stages are typically characterized by elevated levels of amino acids, nucleotides, and terpenoid compounds—metabolites closely associated with both flavor intensity and therapeutic potential (Harada et al., 2003; Son et al., 2019). As maturation progresses, however, these profiles often shift toward carbohydrate accumulation and energy storage, subtly altering both taste and nutritional value. Similar developmental trends have been observed across plant systems, reinforcing the broader biological principle that metabolic states are tightly linked to growth phases (Lee et al., 2015; Jang et al., 2015).
The mechanisms underlying these transitions are increasingly being unraveled through integrated “omics” approaches. Metabolomics, when combined with transcriptomics and proteomics, allows for a more holistic understanding of how metabolic networks are regulated. Studies have shown that gene-to-metabolite interactions form intricate networks that ultimately determine the biochemical output of an organism (Hirai et al., 2005; Wang et al., 2013). In mushrooms, these tools have revealed how environmental factors—such as nitrogen availability or cultivation method—can modulate metabolite accumulation, influencing both sensory properties and bioactivity (Kim et al., 2016; Huang et al., 2018).
Yet, despite these advances, our understanding of fungal metabolomes remains incomplete. The annotation of metabolites, particularly secondary compounds, continues to lag behind data generation, leaving a significant portion of biochemical diversity unexplored (Viant et al., 2017). This gap is not merely academic; it has practical implications for food science, pharmacology, and biotechnology, where unidentified compounds may hold considerable functional potential.
At the same time, the beneficial narrative surrounding mushrooms is counterbalanced by the persistent threat posed by toxic species. Among these, Amanita phalloides, commonly known as the Death Cap, stands out as one of the most notorious. Its toxicity is primarily attributed to amatoxins—cyclic peptides that selectively inhibit RNA polymerase II, thereby halting protein synthesis and triggering cellular apoptosis (Li & Oberlies, 2005; Walton, 2018). Clinically, poisoning follows a deceptive progression, beginning with a latent phase before advancing to gastrointestinal distress and, ultimately, acute liver failure (Ye & Liu, 2018; Kayes & Ho, 2024).
What makes A. phalloides particularly dangerous is not only its biochemical potency but also its morphological similarity to edible species. Misidentification remains a recurring issue, sometimes with fatal consequences. Historical accounts and toxicological studies alike underscore the enduring challenge of distinguishing safe from hazardous fungi (Marmion & Wiedemann, 2002; Ye & Liu, 2018). In this context, analytical techniques have become indispensable. Modern methods such as UPLC-MS/MS offer rapid and precise detection of amatoxins, significantly improving diagnostic accuracy and clinical response (Kayes & Ho, 2024).
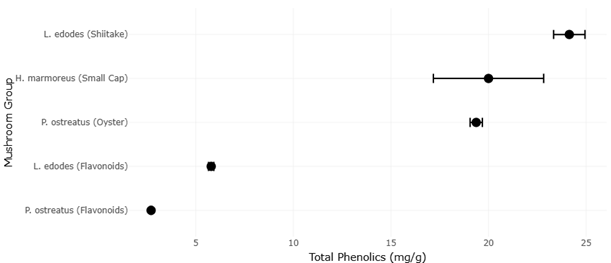
Beyond toxicity, the broader biochemical diversity of mushrooms continues to attract attention for therapeutic applications. Bioactive compounds, including phenolics, flavonoids, and polysaccharides, have been associated with anti-inflammatory, hypoglycemic, and antimicrobial effects (Muszynska et al., 2018; Hwang et al., 2005). Certain species also produce antibacterial substances capable of inhibiting pathogenic microbes, suggesting potential roles in combating antibiotic resistance (Smânia et al., 1995; Rahi & Malik, 2016).
Interestingly, these functional properties are not static but are shaped by environmental and developmental factors. Cultivation strategies, including strain selection and breeding, have been employed to enhance desirable traits such as flavor and bioactivity (Sun et al., 2014; Lee et al., 2014). Enzymatic processes involved in fruiting body formation further illustrate the complexity of fungal development and its influence on metabolite production (Wagemaker et al., 2007; Moore et al., 2008).
Taken together, these observations suggest that mushrooms represent a highly dynamic biological system, where nutritional, therapeutic, and toxicological properties are deeply interconnected. The same metabolic pathways that generate beneficial compounds can, under different circumstances, produce potent toxins. Understanding this balance requires not only detailed biochemical analysis but also an integrative perspective that considers ecological, developmental, and technological factors.
Against this backdrop, the present review seeks to synthesize current knowledge on edible and poisonous mushrooms. By integrating findings from metabolomics, proteomics, and toxicology, the review aims to provide a comprehensive framework for understanding fungal bioactivity. Particular emphasis is placed on how metabolite composition varies across species, developmental stages, and environmental conditions, and how these variations translate into functional outcomes. Ultimately, such an approach may help bridge the gap between traditional knowledge and modern scientific insight, guiding both safe consumption and innovative applications in food and medicine.